Building blocks of matter g3
- 1. 1
- 2. Ch. 11 , Lesson 6 ( Building blocks of Matter ) What objects do Magnet Attract?? Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 2
- 3. Ch. 11 , Lesson 6 ( Building blocks of Matter ) • Magnet attract some objects and not others. • Objects magnet attracts are made of certain metals. • Is hard, shiny material found in Earth’s ground. • Magnets attract metal called iron, and some other metals too. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 3 Metal
- 4. Ch. 11 , Lesson 6 ( Building blocks of Matter ) • Anything attracts metals has the property of magnetism. • Magnetism is very useful property. How?? • It holds things • It is used to sort certain metals from other objects. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 4
- 5. Ch. 11 , Lesson 6 ( Building blocks of Matter ) Do you need iron in your body? Yes, you need it every day to stay healthy. From what can you get iron? From meat and dark green vegetables like spinach. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 5
- 6. Ch. 11 , Lesson 6 ( Building blocks of Matter ) • Iron is often mixed with other materials to make steel. • Steel is strong and lasts a long time. • It is used to build things like bridges, railroads, and cars. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 6
- 7. Ch. 11 , Lesson 6 ( Building blocks of Matter ) Other metals such as: gold, silver, copper, and aluminum. Each metal has its own properties. Copper and Aluminum soft and light. Copper used to make pipes. Aluminum used to make airplanes. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 7
- 8. Ch. 11 , Lesson 6 ( Building blocks of Matter ) • Gold is soft, yellow, and shiny • Silver shiny too. • They are used to make jewelry. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 8
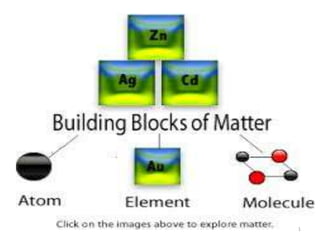
- 9. Ch. 11 , Lesson 6 ( Building blocks of Matter ) What are the building blocks of matter? the metals iron, gold, silver and copper are elements. the building blocks of matter. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 9 Elements
- 10. Ch. 11 , Lesson 6 ( Building blocks of Matter ) • There are more than 100 different elements. • They make up all the matter in the world. • Some elements such as iron and copper are solids • Other elements such as mercury are liquids • Others such as helium gases • Each element has its own properties. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 10
- 11. Ch. 11 , Lesson 6 ( Building blocks of Matter ) • Elements join together in different ways . • They form everything on Earth. • All elements are made of atoms. • Is the smallest particle of matter. • It is too small to see with your eyes. • The atoms that make up one element are all alike. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 11 Atom
- 12. Ch. 11 , Lesson 6 ( Building blocks of Matter ) • Long ago people thought the only elements were air, water, fire and Earth. • But today they understand they are not elements. • The tusks of the a walrus contain calcium, which is element. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 12
- 13. Ch. 11 , Lesson 6 ( Building blocks of Matter ) • Water can break apart into 2 gases( hydrogen + oxygen) both of these gases are elements. • H2O = 2Hydrogen + 1 Oxygen • Wood when heated, it gives off carbon gases, so carbon is element but wood isn’t. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 13
- 14. Ch. 11 , Lesson 6 ( Building blocks of Matter ) • The periodic table • It lists all the elements. • The letters in each box stand for the name of the elements. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 14
- 15. Ch. 11 , Lesson 6 ( Building blocks of Matter ) What happens when Elements join together? How 100 elements form all materials on Earth? Elements join together to form compounds. Forms when you put 2 or more elements together. They have different properties from the elements they are made of. Ex. Rust = iron + oxygen Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 15 Compound
- 16. Ch. 11 , Lesson 6 ( Building blocks of Matter ) Common Compounds Salt Rust Wood Sodium + chloride Iron + oxygen Carbon + oxygen +hydrogen Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 16
- 17. Ch. 11 , Lesson 6 ( Building blocks of Matter ) • When compound forms it is chemical change. • Changes in the matter itself. • You start with one kind and end with another. • Ex. Burning wood ash+ gases • Rust is very different from iron • Cooking • Your body uses food for its energy. Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 17 Chemical change
- 18. Ch. 11 , Lesson 6 ( Building blocks of Matter ) Vocabulary: Metal Element Atom Compound Chemical change Objectives: I will be able to know: 1-what is magnet and its properties. 2-elements are the building blocks of matter. 3-differentiate between physical and chemical change. 18
- 19. 19
- 20. 20 Physical Change Chemical Change Folding Tearing Change state Burning Rusting cooking Matter looks different but still the same kind New material forming Color changes Chemical properties change