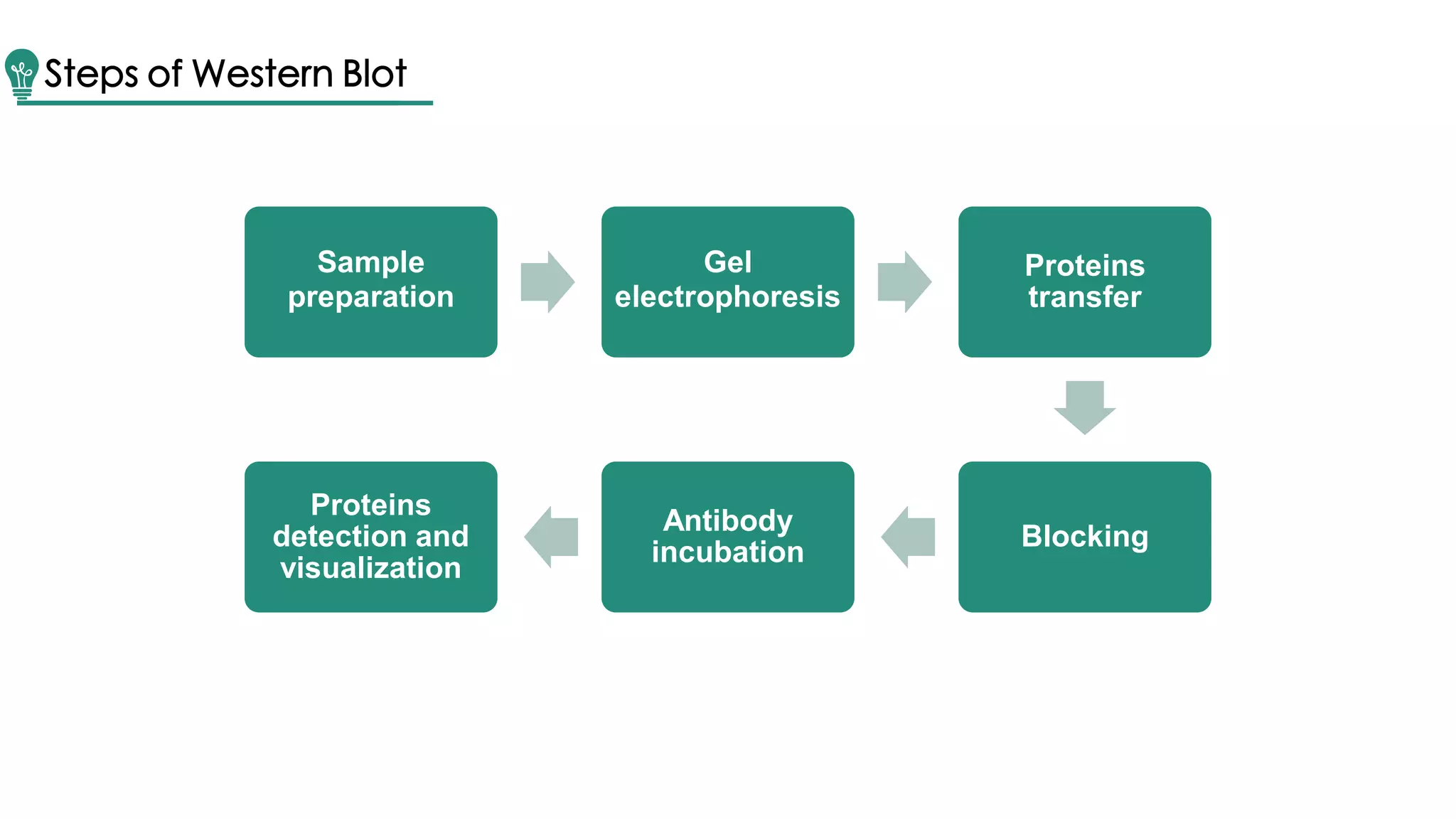
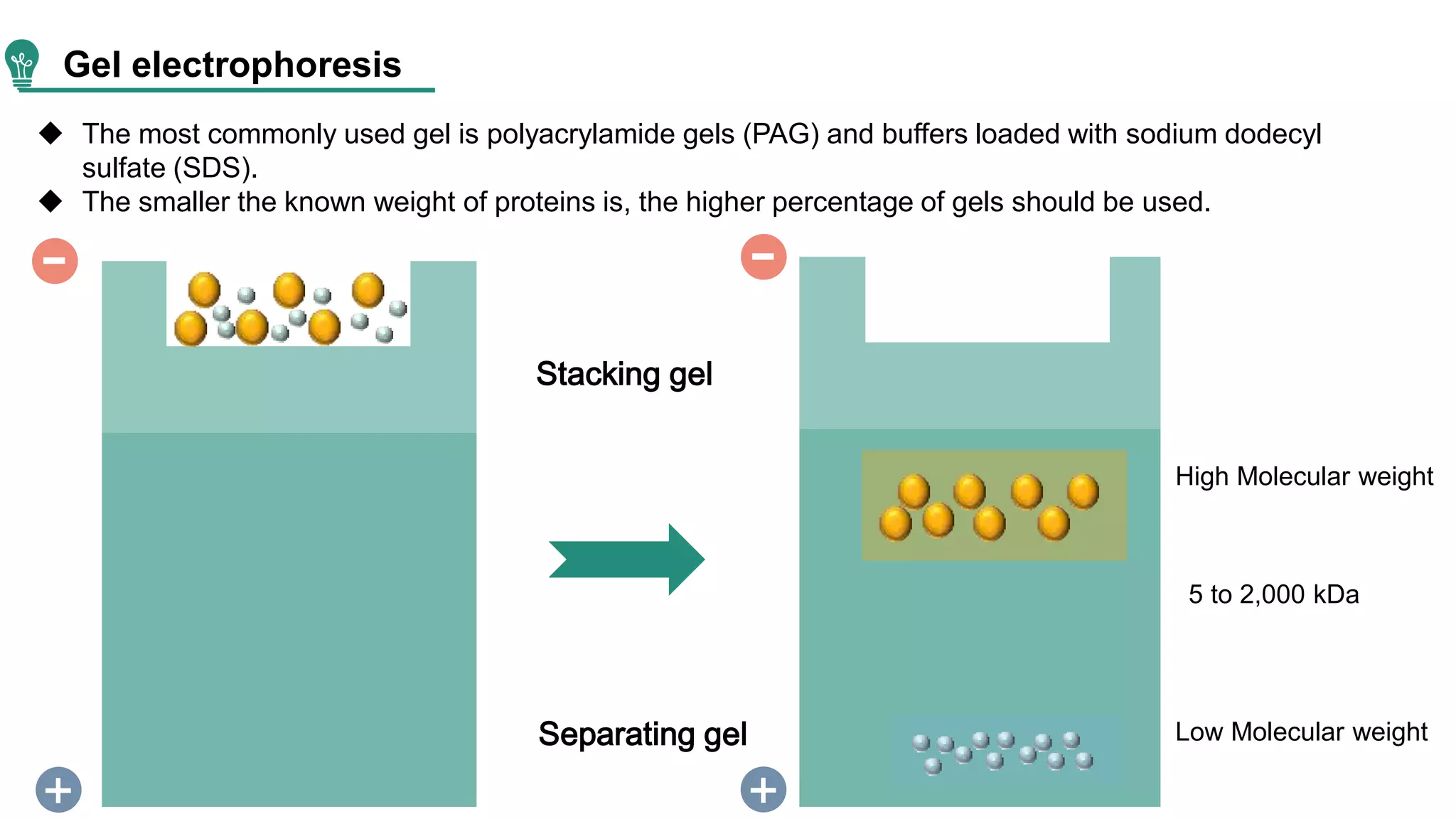
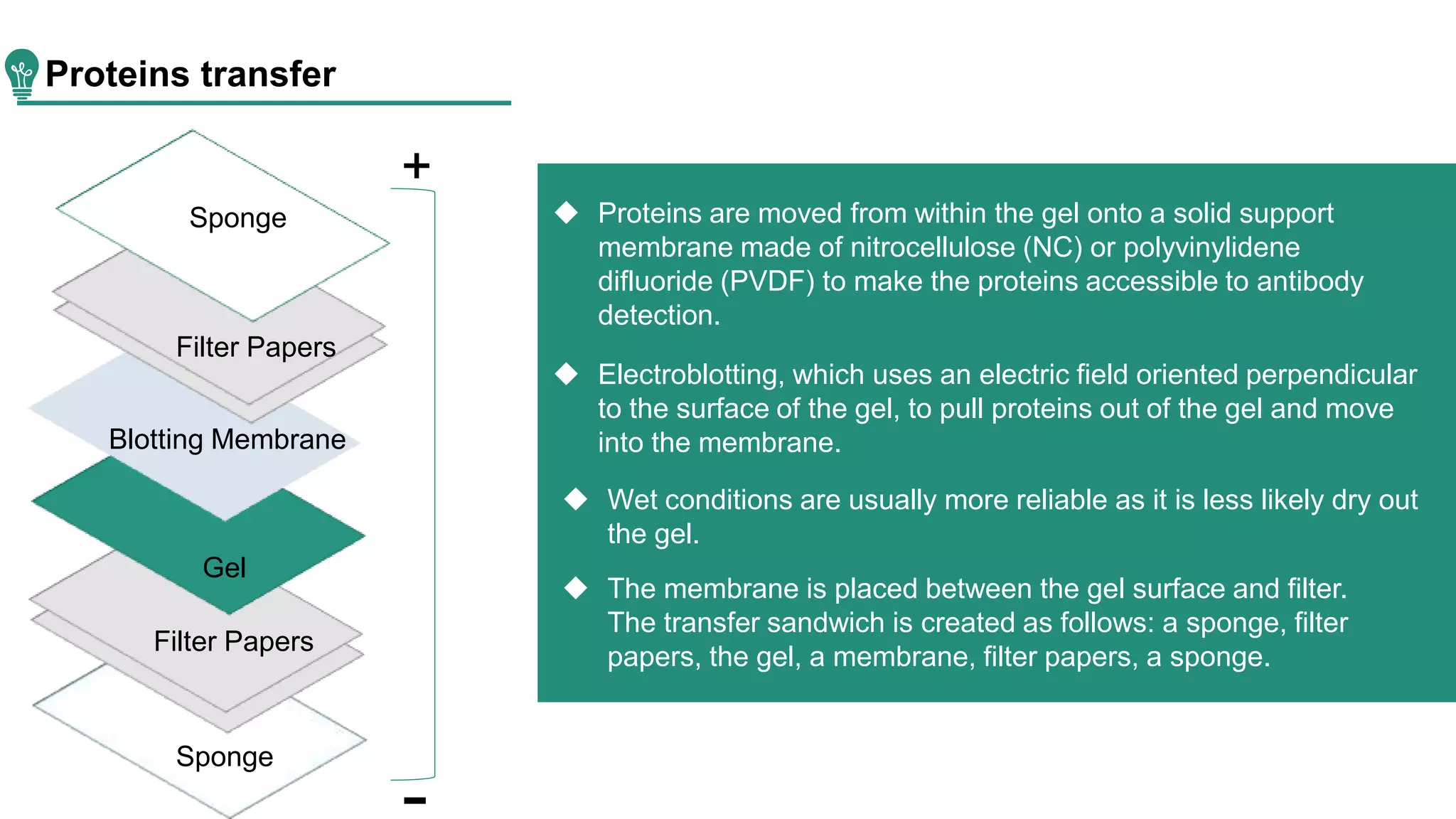
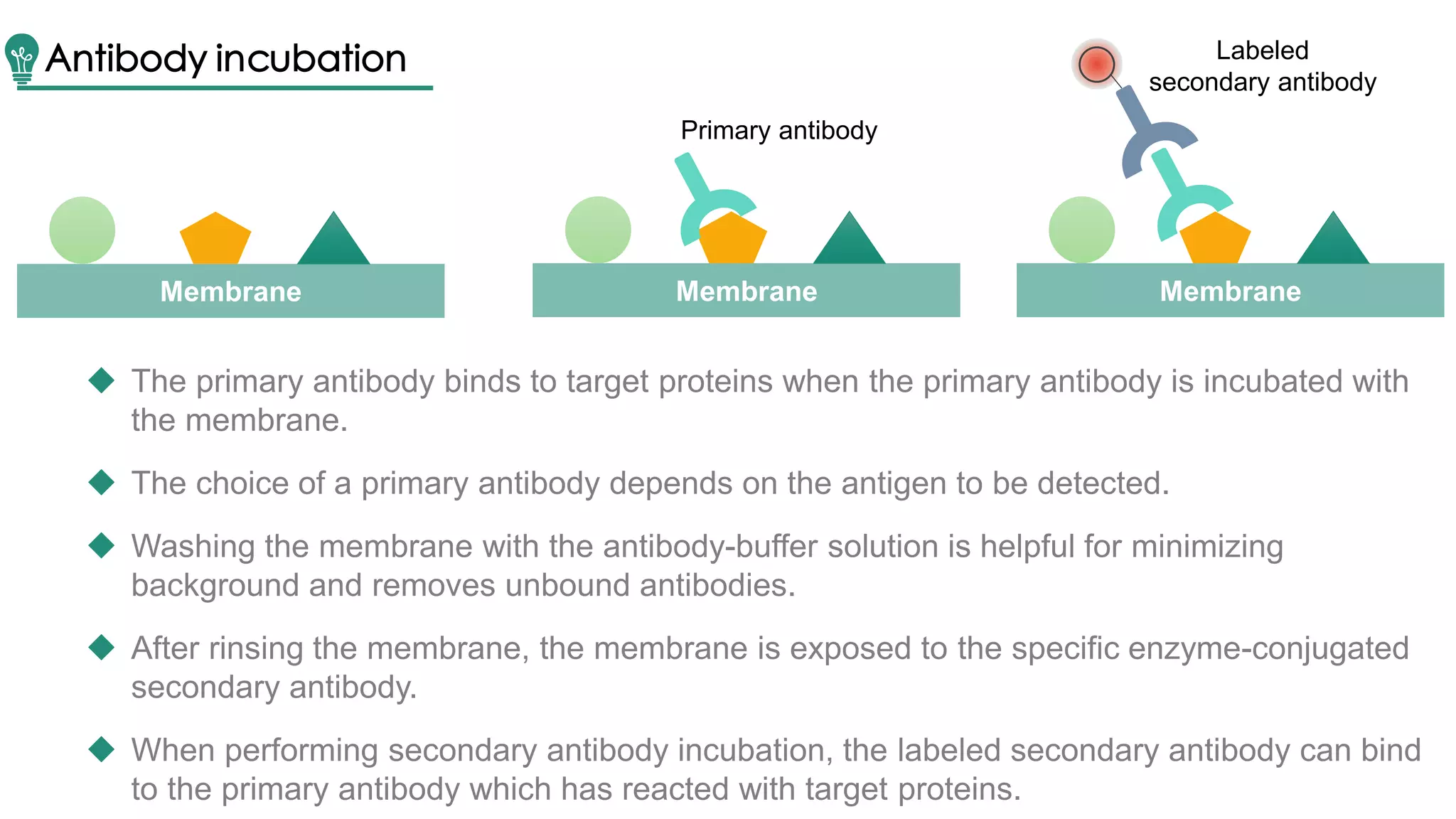
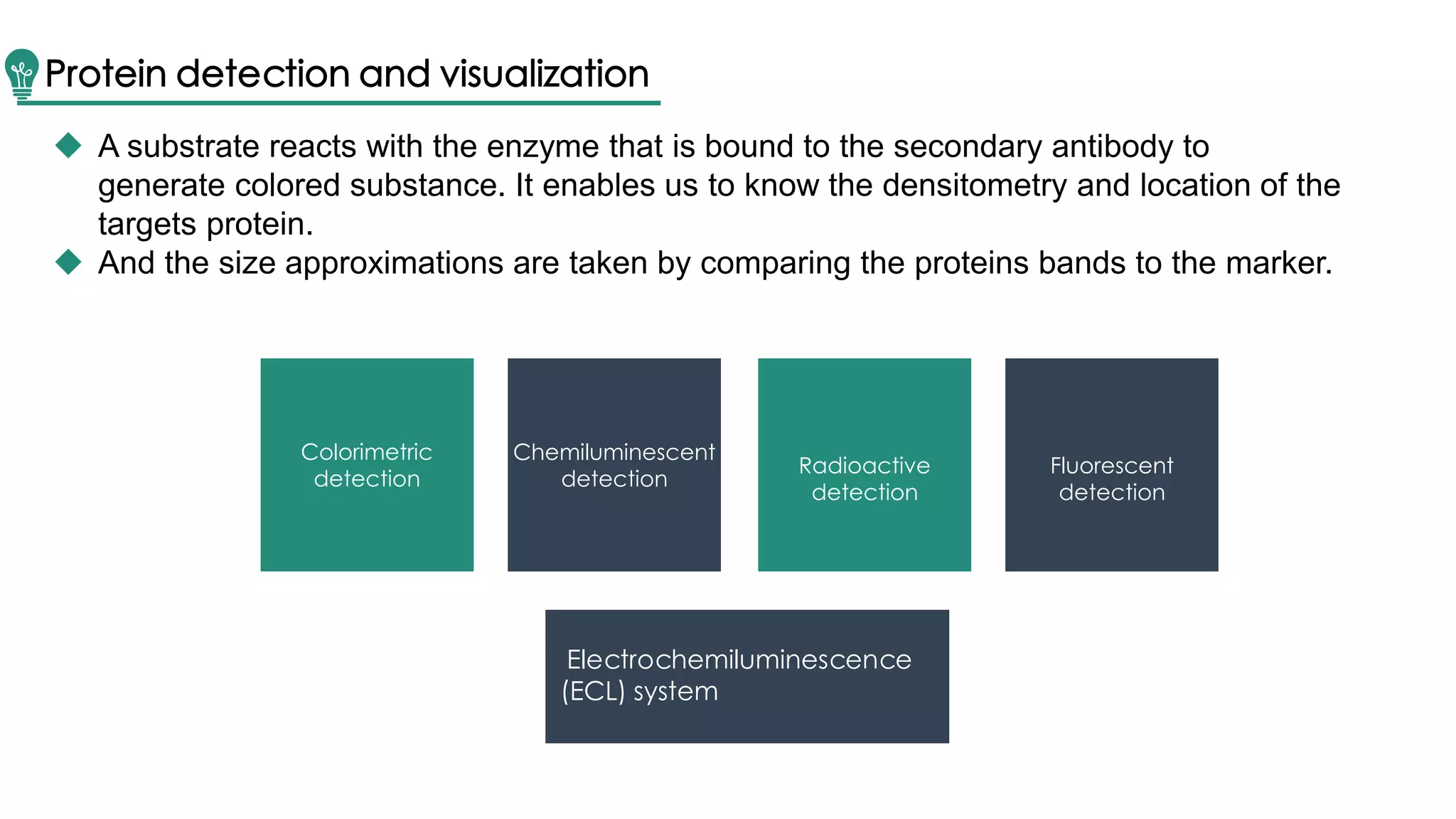
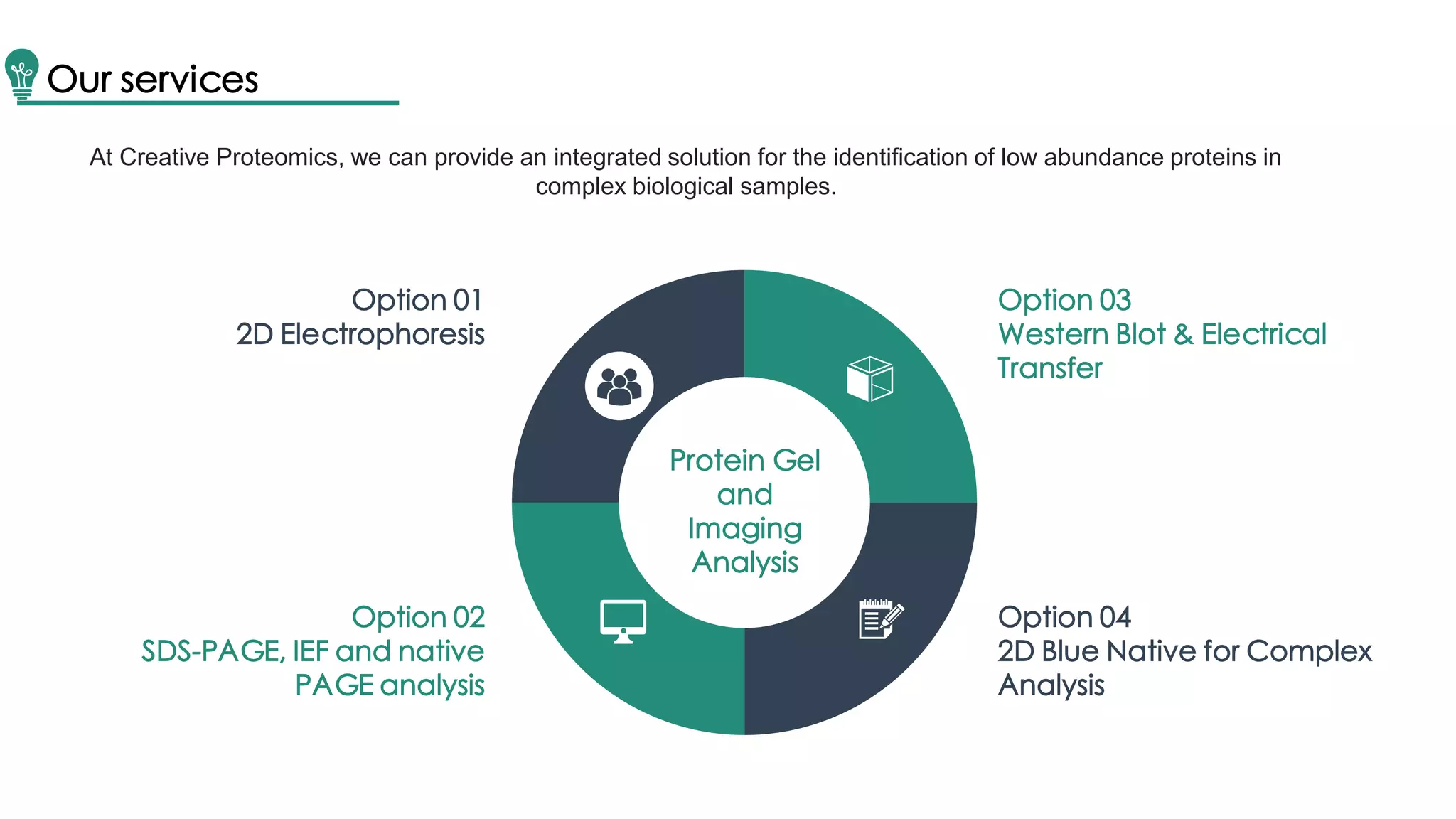
Western blot is a method for protein analysis, introduced in 1979, that allows for qualitative and semi-quantitative assessments by separating proteins by size, transferring them to a solid support, and using antibodies for visualization. The process includes sample preparation, gel electrophoresis, transferring proteins to membranes, blocking, antibody incubation, and detection through various methods. Creative Proteomics offers services for identifying low abundance proteins in complex biological samples.