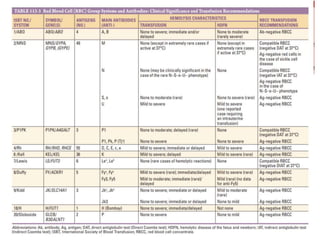
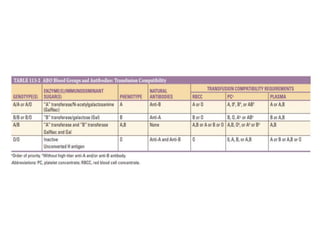
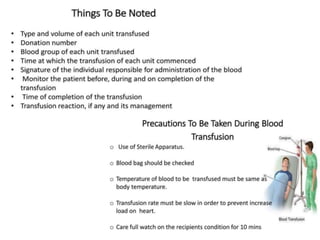
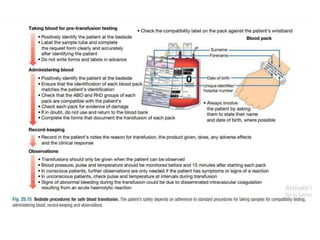
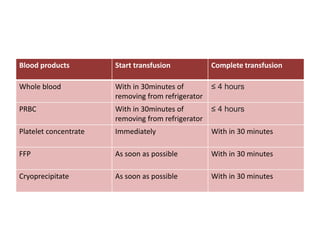
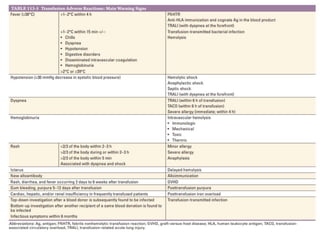
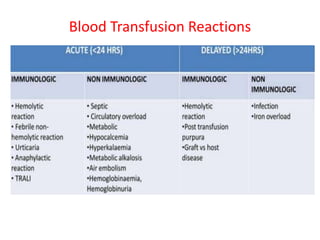
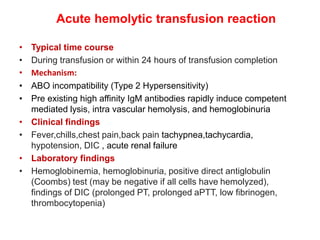
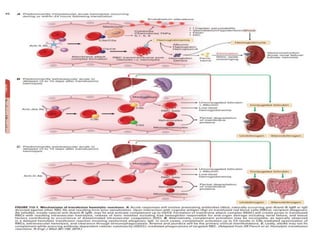
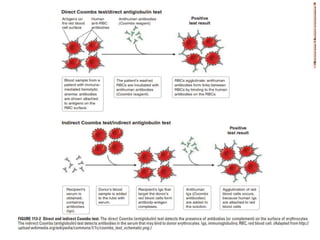
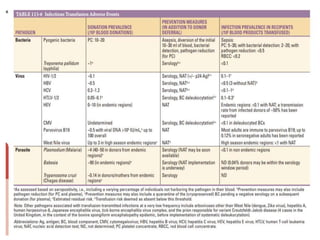
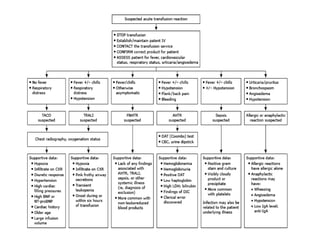
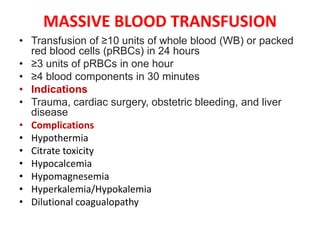
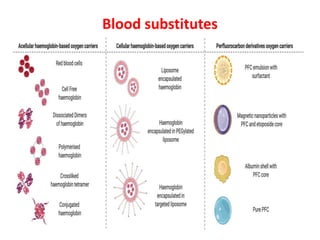
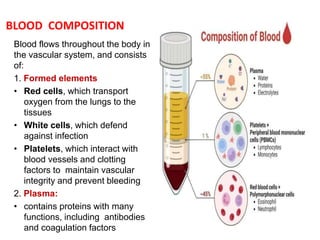
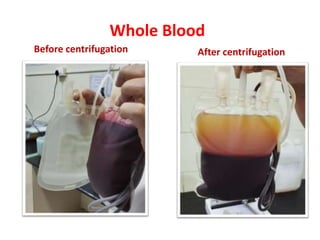
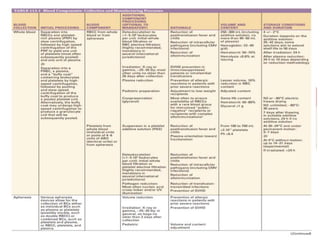
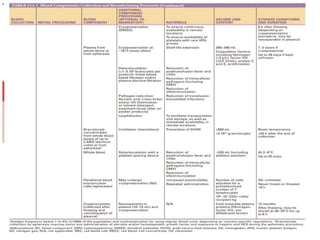
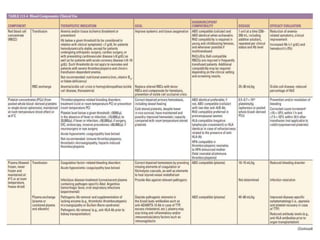
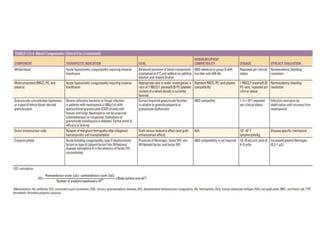
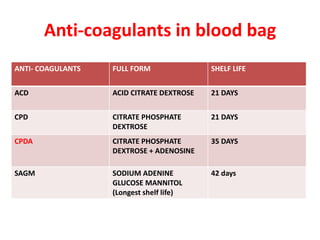
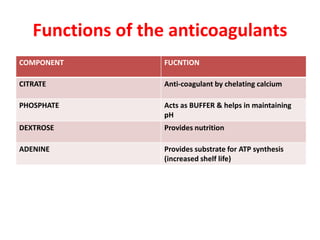
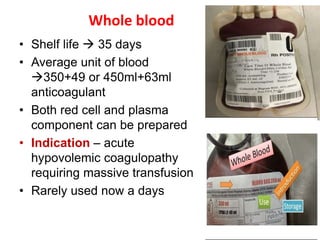
This document discusses blood and blood components. It describes the composition of blood and its formed elements and plasma. It then discusses the various blood components that can be prepared including packed red blood cells, platelets, fresh frozen plasma, cryoprecipitate, and describes the processes involved in collecting and preparing these components. It also covers topics like shelf life, dosing, and indications for transfusion of the various components. It discusses some potential transfusion reactions like acute hemolytic, delayed hemolytic, febrile non-hemolytic, sepsis/bacterial infection, and allergic reactions.




![• Irradiation is used to prevent ta-GVHD in at-risk individuals
• Patients at most risk include those who are severely
immunocompromised and those who would fail to recognize the
transfused lymphocytes as foreign, due to human leukocyte antigen
(HLA) homology
• Indications
• Recipients of intrauterine or neonatal exchange transfusion;
premature neonates
• Individuals with congenital cell-mediated immunodeficiency states
• Individuals treated with specific types of potent immunosuppressive
therapies (purine analogs, antithymocyte globulin [ATG], certain
monoclonal antibodies); this may include those being treated for
non-Hodgkin lymphoma (NHL) or other hematologic malignancies
• Recipients of hematopoietic stem cell transplant (autologous or
allogeneic)
• Individuals with Hodgkin lymphoma (any stage of disease)
• Individuals at risk for partial HLA matching with the donor due to
directed donations, HLA-matched products, or genetically
homogeneous populations](https://image.slidesharecdn.com/bloodandbloodcomponents-231130065426-d4160035/85/blood-and-blood-components-pptx-14-320.jpg)