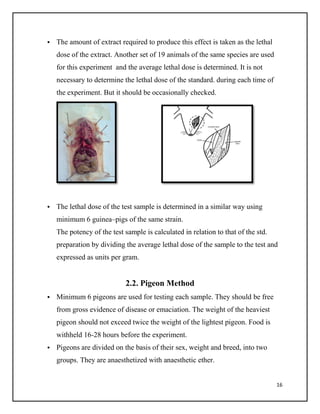
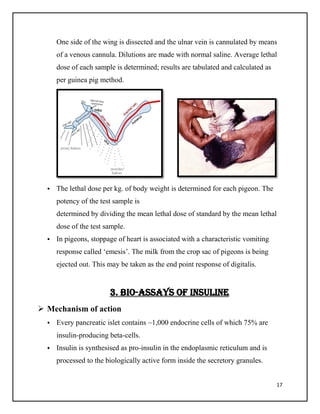
This document summarizes biological assays, specifically focusing on pharmaceutical quality management. It defines an assay as a procedure used to qualitatively or quantitatively assess the presence, amount, or functional activity of an analyte, such as a drug or biochemical substance. There are three main types of assays discussed: chemical assays, immunoassays, and bioassays. Bioassays are used to estimate the concentration or potency of a pharmaceutical drug by comparing its effects on animals or humans to a standard. The document outlines various techniques for bioassays including quantal, graded, and effect over time assays. It also discusses standards, accuracy, dependencies on dose and intensity, and applications of bioassays in pharmaceutical quality management.