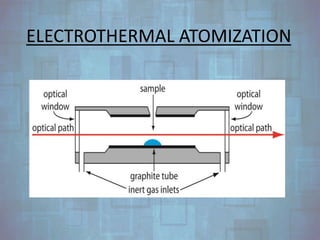
Atomic absorption spectroscopy is a technique that uses the absorption of light to measure the concentration of atomic absorption in a sample. It works by vaporizing the sample into atoms, then measuring how much light is absorbed by the atoms at a specific wavelength. The amount of absorption is directly related to the concentration of the element being measured. The key components of an atomic absorption spectroscopy system are the lamp sources, which emit specific wavelengths of light corresponding to the element being analyzed, the atomization process which turns solid or liquid samples into gaseous atoms, detectors that convert light signals into electrical signals, and monochromators that isolate the desired wavelength of light. Potential sources of interference must also be considered and addressed. Common applications of atomic absorption spectroscopy