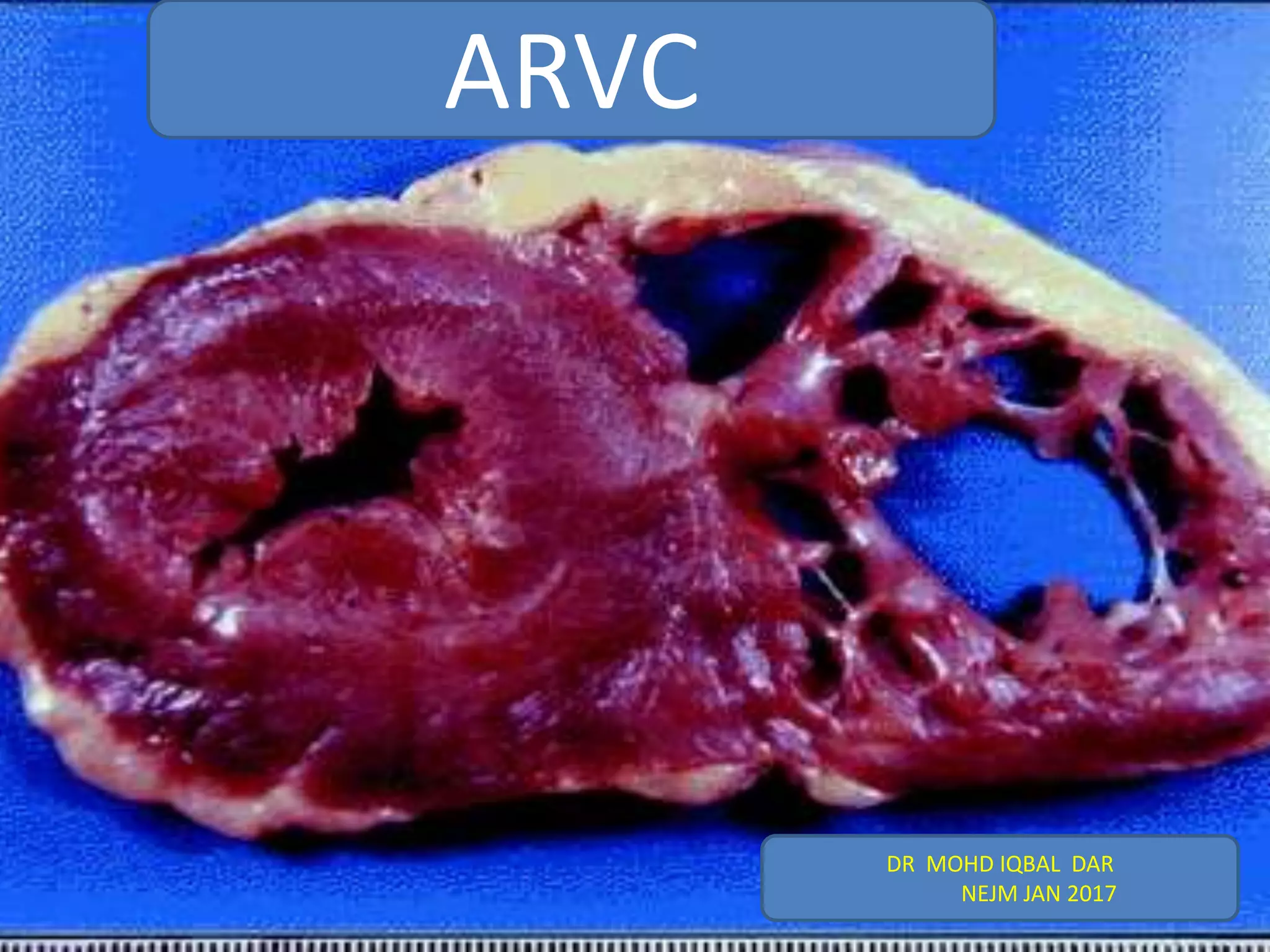
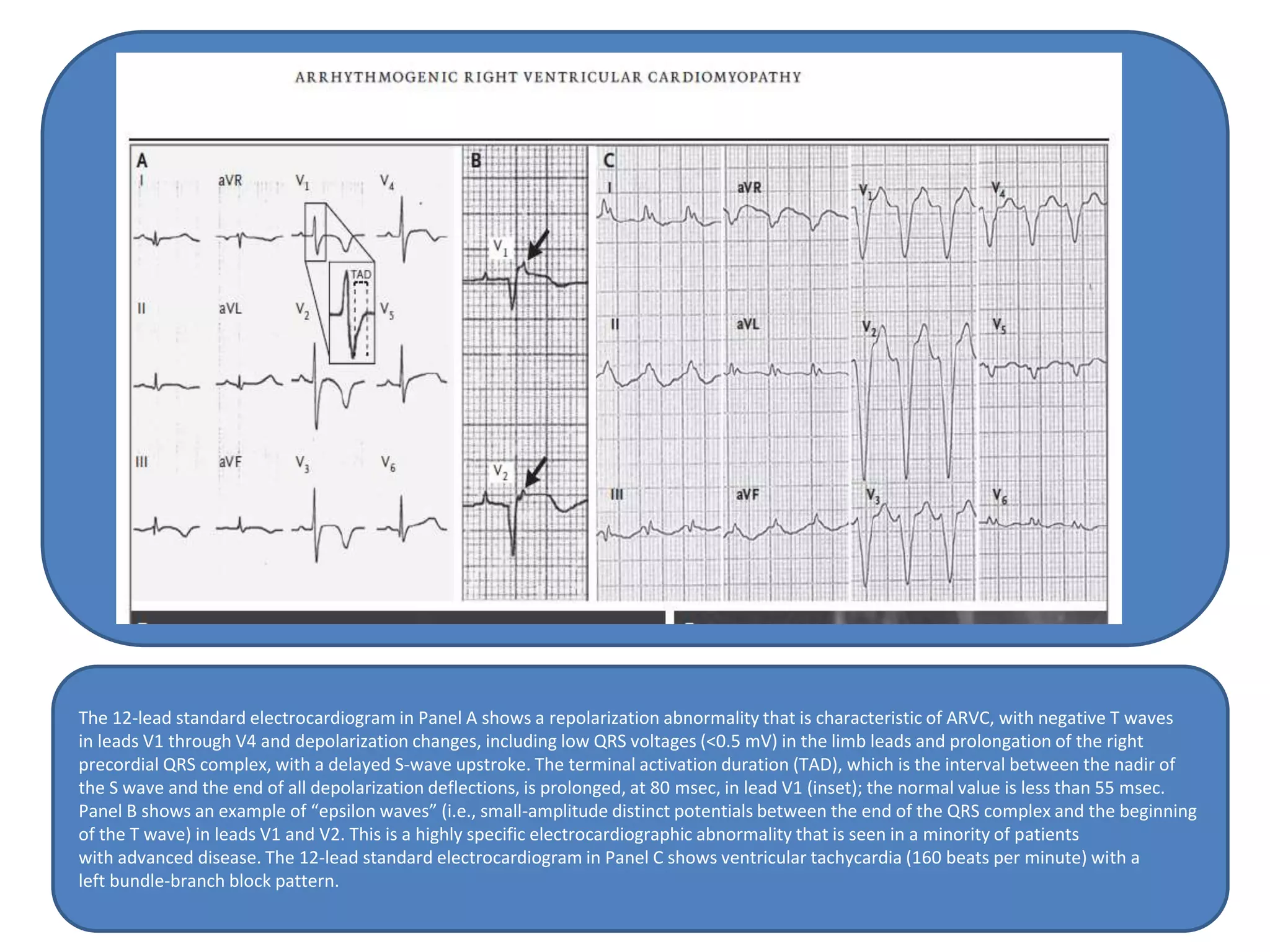
ARVC is a heritable heart muscle disorder that predominantly affects the right ventricle. It is caused by genetic defects in cardiac desmosomes, which are important for cell-to-cell adhesion. This leads to progressive loss of right ventricular myocardium and replacement by fibrofatty tissue. ARVC can cause dangerous ventricular arrhythmias and is a leading cause of sudden cardiac death in young people. Diagnosis involves imaging tests and electrocardiography to detect right ventricular structural abnormalities and arrhythmias.