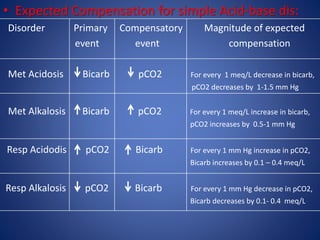
The document provides a 6-step approach to analyzing arterial blood gases (ABGs). The 6 steps are: 1) analyze pH, 2) analyze pCO2, 3) analyze HCO3, 4) match CO2 or HCO3 to pH, 5) check for opposite direction of compensation, and 6) analyze pO2 and oxygen saturation. Examples are given of how to use the 6 steps to diagnose different acid-base disturbances such as respiratory acidosis, metabolic alkalosis, and mixed disorders. Normal values and concepts like the anion gap and types of respiratory failure are also explained.













![• Appropriate Compensation During Simple Acid-
Base Disorders:
• DISORDER EXPECTED COMPENSATION
• Metabolic acidosis PCO2 = 1.5 × [HCO3
-] + 8 ± 2
• Metabolic alkalosis For each 10 mEq/L increase in serum
[HCO3
-] , PCO2 increases by 7 mm Hg .](https://image.slidesharecdn.com/abcofabg-160305175440/85/ABC-of-ABG-Dr-Padmesh-18-320.jpg)
![• Appropriate Compensation During Simple Acid-
Base Disorders:
• DISORDER EXPECTED COMPENSATION
• Respiratory acidosis
• Acute : For each 10-mm Hg increase in PCO2 , [HCO3
-] increases
by 1.
• Chronic : For each 10-mm Hg increase in PCO2 , [HCO3
-]
increases by 3.5 .
• Respiratory alkalosis
• Acute: For each 10mm Hg decrease in PCO2 , [HCO3
-] falls by 2.
• Chronic : For each 10mm Hg decrease in PCO2,[HCO3
-] falls by 4](https://image.slidesharecdn.com/abcofabg-160305175440/85/ABC-of-ABG-Dr-Padmesh-19-320.jpg)





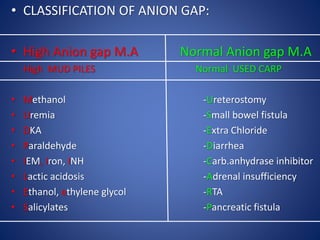
![• ANION GAP:
• Anion gap indicates the presence or absence of increased levels of
unmeasured anions (proteins, phosphates, SO4, organic anions)
(Na + K + Ca + Mg + unmeasured cations) = (Cl + HCO3 + unmeasured anions)
AG = Measured Cations – Measured Anions
• AG =[ Na – (Cl + HCO3) ] = [ 140 - (104+24) ] = 12 meq/L
• AG =[ (Na + K ) – (Cl + HCO3) ] = [ (140 + 4) - (104+24) ] = 16 meq/L](https://image.slidesharecdn.com/abcofabg-160305175440/85/ABC-of-ABG-Dr-Padmesh-25-320.jpg)
![• ANION GAP:
• Anion gap indicates the presence or absence of increased levels of
unmeasured anions (proteins, phosphates, SO4, organic anions)
(Na + K + Ca + Mg + unmeasured cations) = (Cl + HCO3 + unmeasured anions)
AG = Measured Cations – Measured Anions
• AG = [ Na – (Cl + HCO3) ] = 8 - 12 meq/L
• AG = [ (Na + K ) – (Cl + HCO3) ] = 12 - 20 meq/L](https://image.slidesharecdn.com/abcofabg-160305175440/85/ABC-of-ABG-Dr-Padmesh-26-320.jpg)
![• ANION GAP:
- Increased Anion Gap Metabolic Acidosis:
-Addition of acid load Increase in unmeasured anions
- Normal Anion Gap Metabolic Acidosis:
-Loss of bicarbonate.
-However, this loss of bicarbonate is compensated by
corresponding increase in chloride,so that the anion gap
remains unchanged.
AG = [ Na – ( Cl + HCO3 ) ]
(Hyperchloremic Met.Acidosis)](https://image.slidesharecdn.com/abcofabg-160305175440/85/ABC-of-ABG-Dr-Padmesh-27-320.jpg)