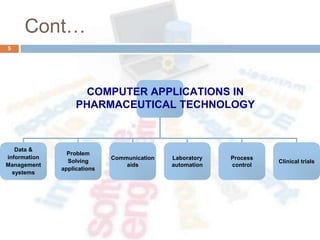
This document outlines the importance of computer system validation (CSV) in the pharmaceutical industry, emphasizing the need for proper guidelines and methodologies to ensure compliance with regulatory requirements. It covers aspects of software and hardware validation, management information systems, and the reconciliation of operational needs with technology. The summary also highlights the specific principles for validating electronic spreadsheets and the significance of documentation in the validation process.