Week 1
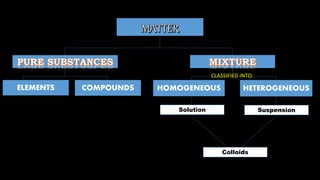
- 1. HOMOGENEOUS HETEROGENEOUSELEMENTS COMPOUNDS Solution Suspension Colloids CLASSIFIED INTO:
- 3. Formed when two or more substances – whether in solid, liquid, or gas forms – are combined. It is classified into two: – the mixture has a uniform appearance, you cannot see the components through our naked eyes. – do not have uniform appearance, therefore components are visible to naked eye.
- 4. EXAMPLE: 1. Vinegar (mixture of water, cane juice, sodium, etc.) 2. Orange juice (powder and water) 3. Coffee (coffee powder and water) 4. Prefume 5. Disinfectant alcohol
- 5. EXAMPLE: 1. Fruit and vegetable salad 2. Water and oil 3. Sand, rocks, and water 4. Flour and water (if not evenly whisk)
- 6. It is classified into three types: 1. Solutions 2. Suspensions 3. Colloids
- 7. It is a homogeneous mixture, and usually in liquid form. Has two factors: Solute – substance that is being dissolved. Solvent – dissolves the solute (water as universal solvent). Example: salt solution Salt as the solute, water as the solvent.
- 8. – refers to the amount of solute being dissolved in a given amount of solvent at particular temperature. CLASSIFIED INTO TWO: - solute dissolve easily into the solvent. Example: sugar and salt, and water. – solutes does not dissolve easily to solvent. Example: rocks, sand, rice grains, corn and pebbles, and water.
- 9. - Stirring using spoons can dissolve the solute faster. - it causes the particles of liquid to move faster and farther apart and come in solid particles faster. - solid particles dissolve easily in hot liquid. - example: creamer dissolved faster in hot liquid than cold one.
- 10. - smaller particles dissolved faster in liquid than the bigger particles. - Ex: chicken powder dissolve easily than chicken cube. - it determines whether they form solution. - Example; oil and water, they did not form solution because they have different nature.
- 11. – small amount of solute is dissolve. – large amount of solute is dissolve. – much more solute in the solution than the solvent.
- 12. – dissolving liquid, solid, or gaseous to liquid solvent. Example: brine (salt and water), alcohol (liquid to liquid), Cola (carbon dioxide gas and water) - alcohol is a miscible solution since they combined evenly with each other substances. Oil and water are called immiscible since they are not attracted to each other.
- 13. – made by mixing two or more gases. Example: air (combination of carbon dioxide, oxygen, nitrogen and other gases) – mixing solid with solid, liquid and gas. Example: steel (carbon and iron) brass (zinc and copper)
- 14. to the food such as salt and sugar solution – dissolve grease and stain in clothes (soaps) – tea, coffee, soft drinks – medicine we intake into the blood stream.