Embed presentation
Download to read offline




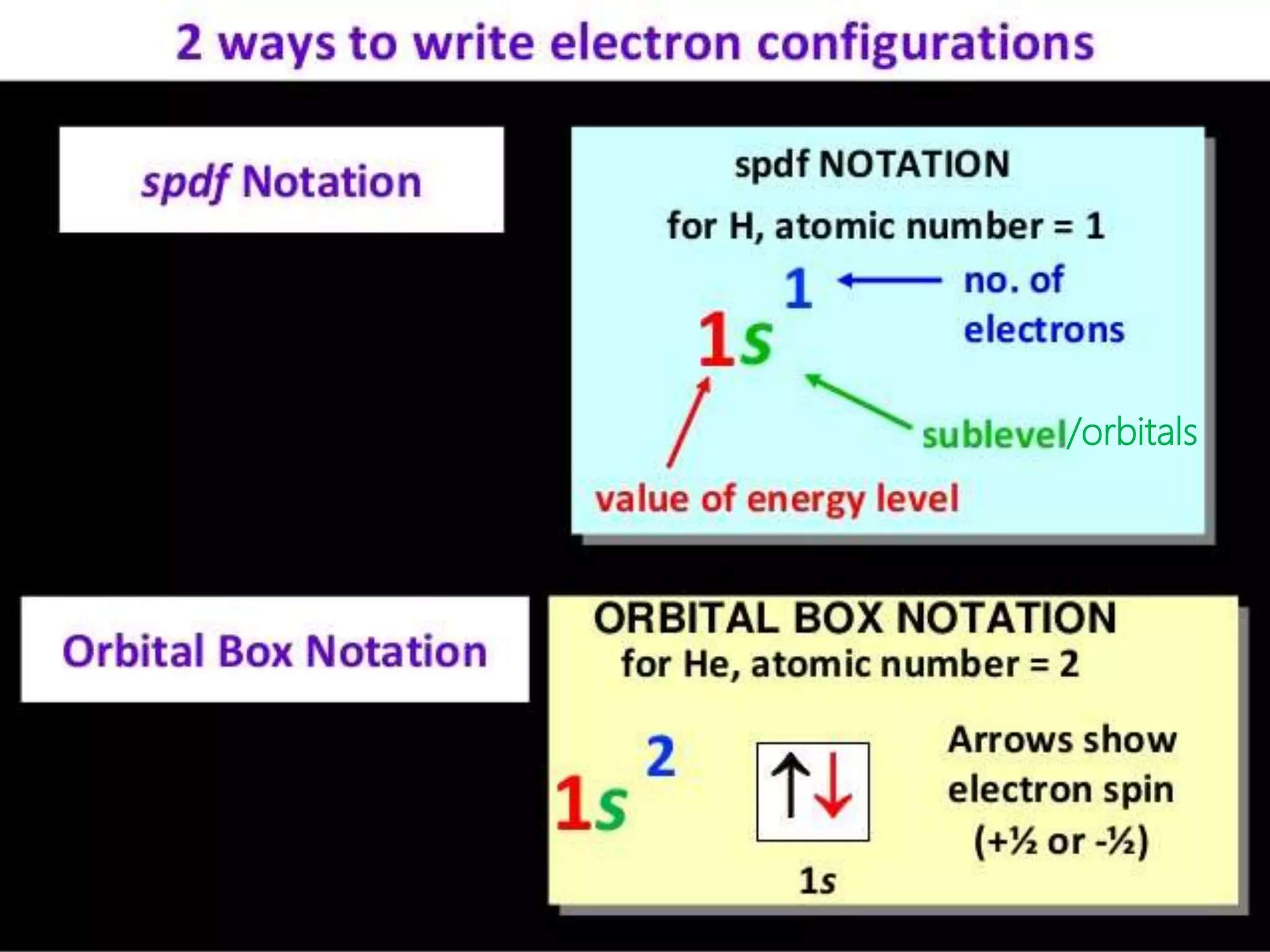
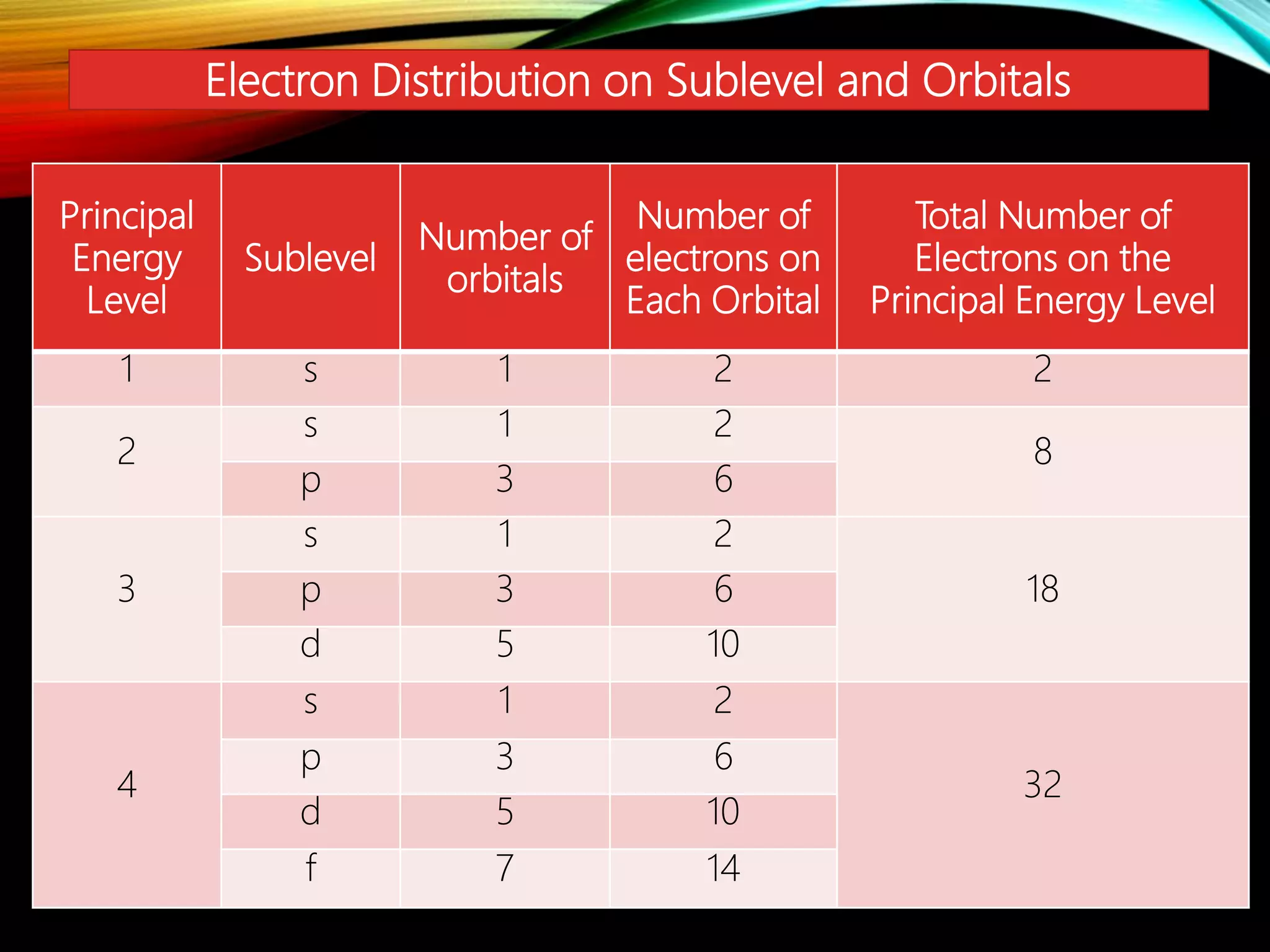





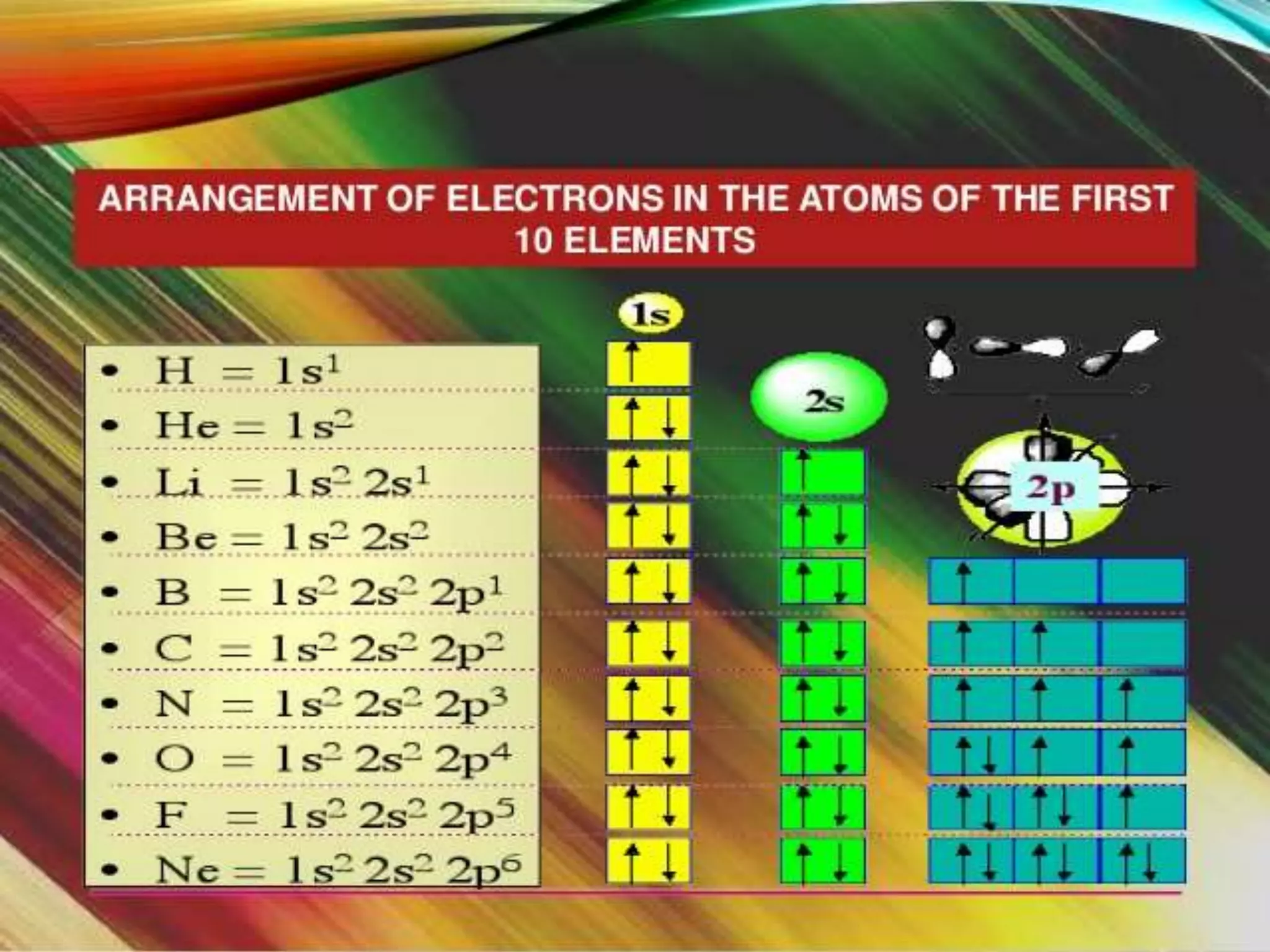

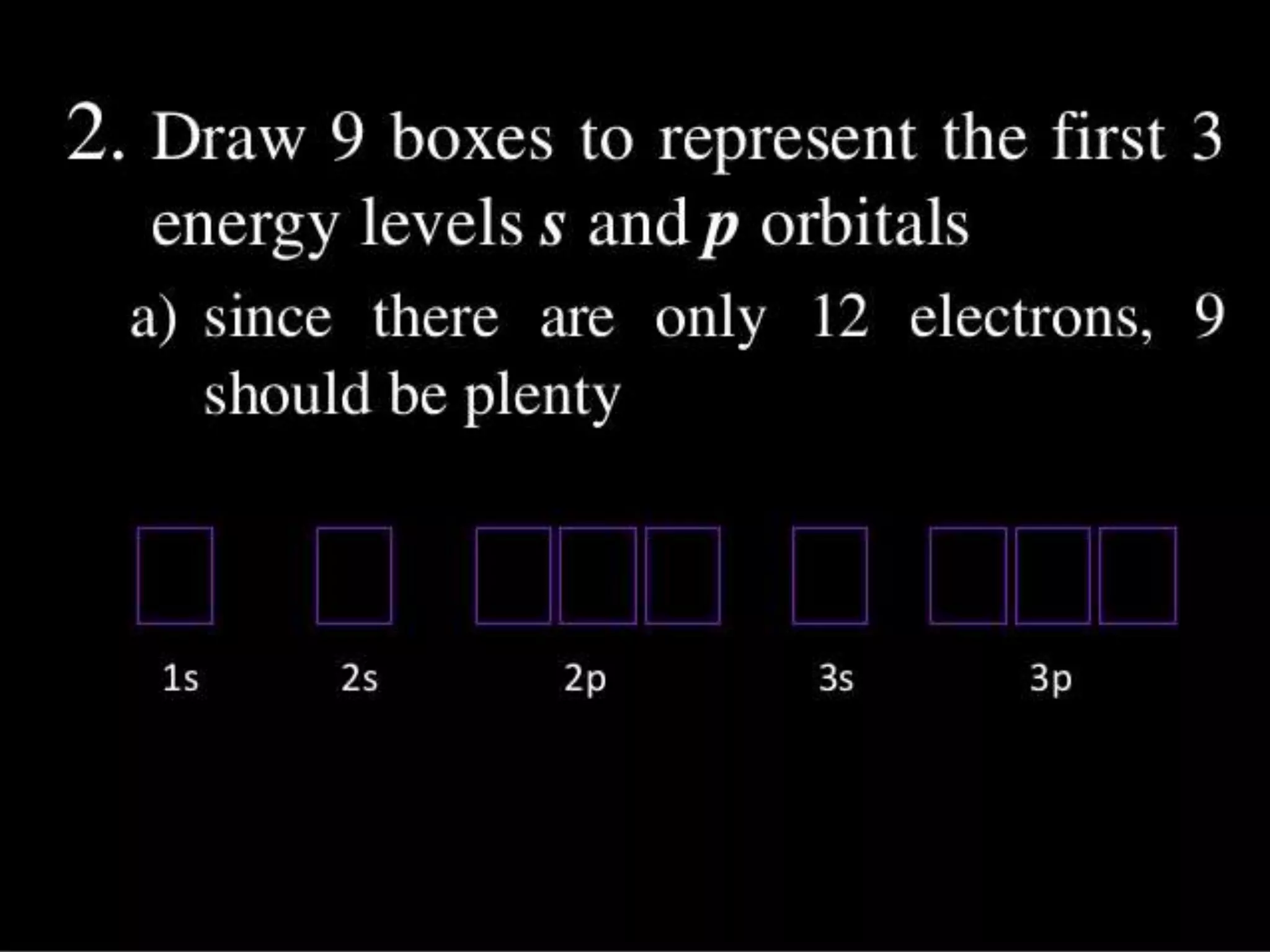


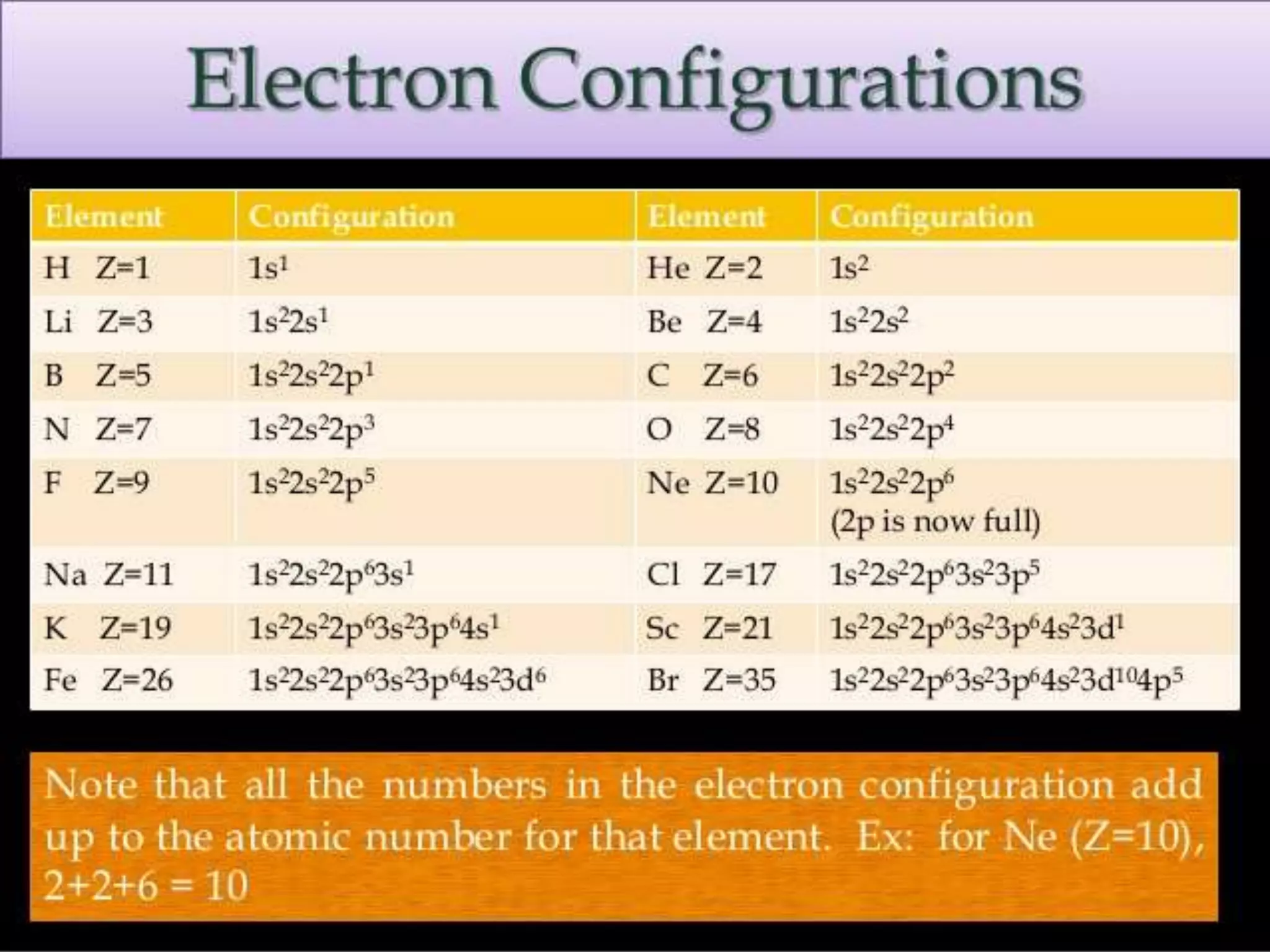









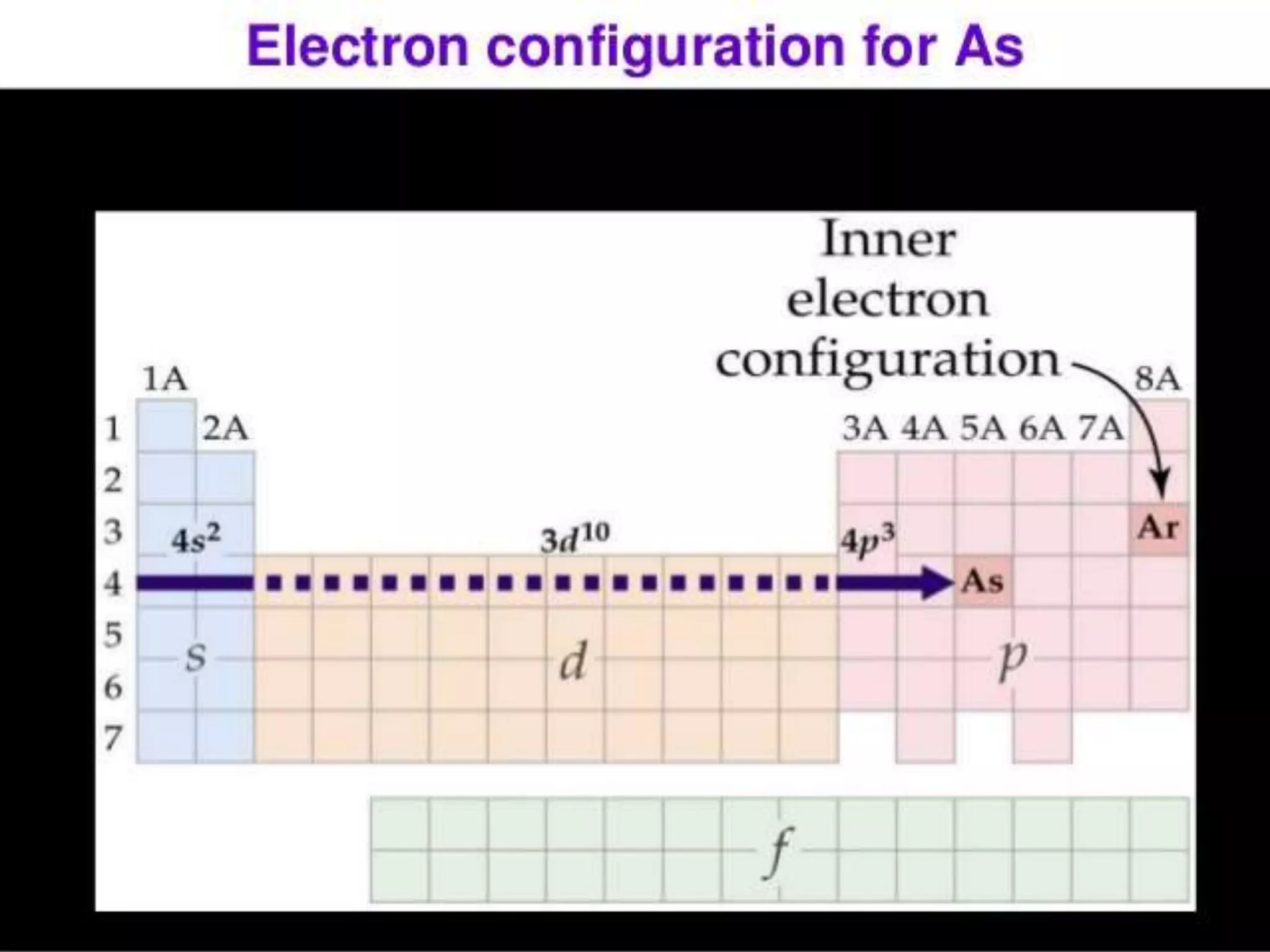




This document discusses the electron distribution in orbitals and sublevels across the first four principal energy levels. It provides the number of orbitals and electrons in each sublevel (s, p, d, f) as well as the total electrons per principal energy level. The first level contains 2 electrons in the s orbital. Each subsequent level adds new sublevels with increasing numbers of orbitals and electrons as you move further from the atomic nucleus.






























