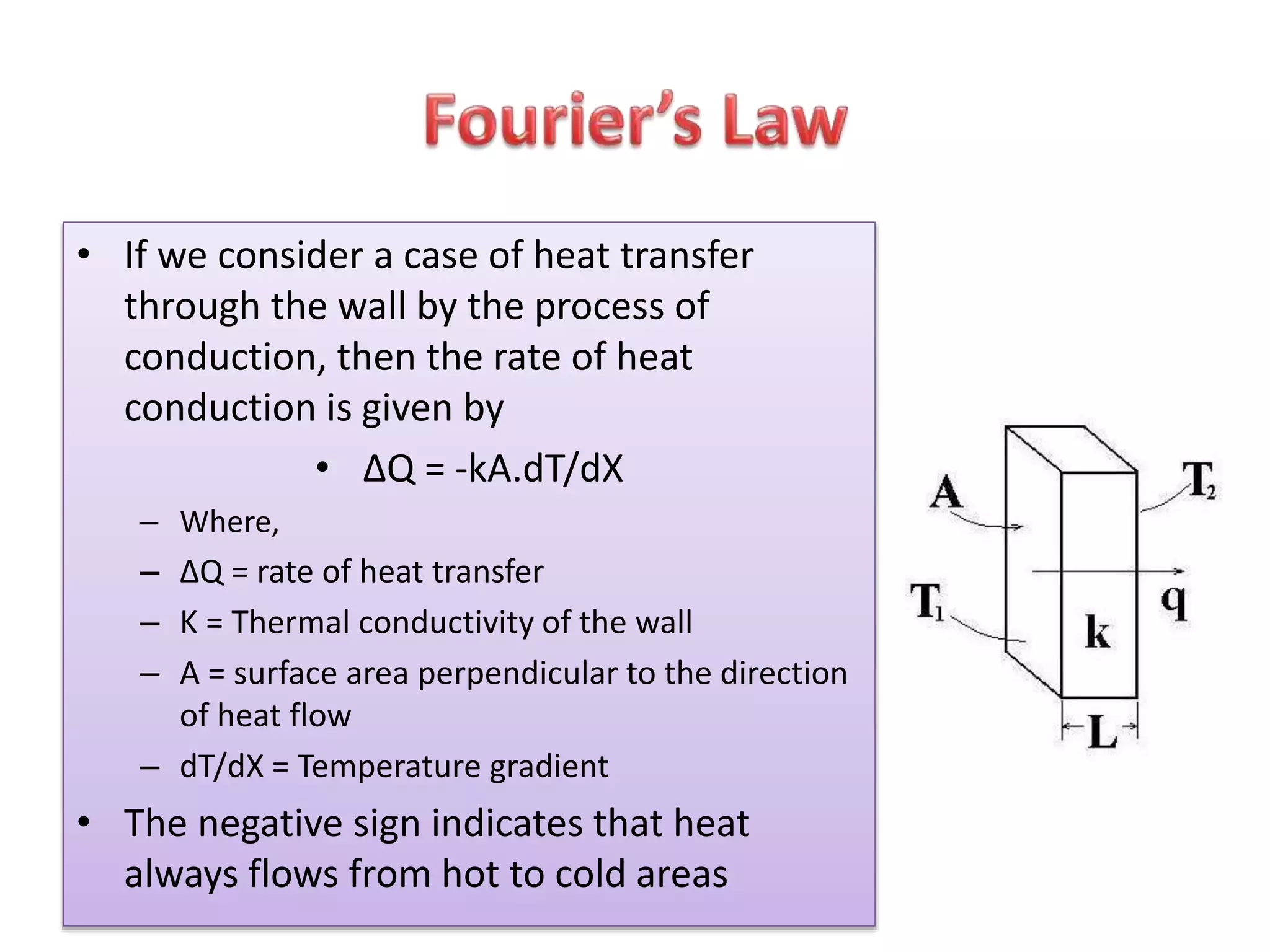
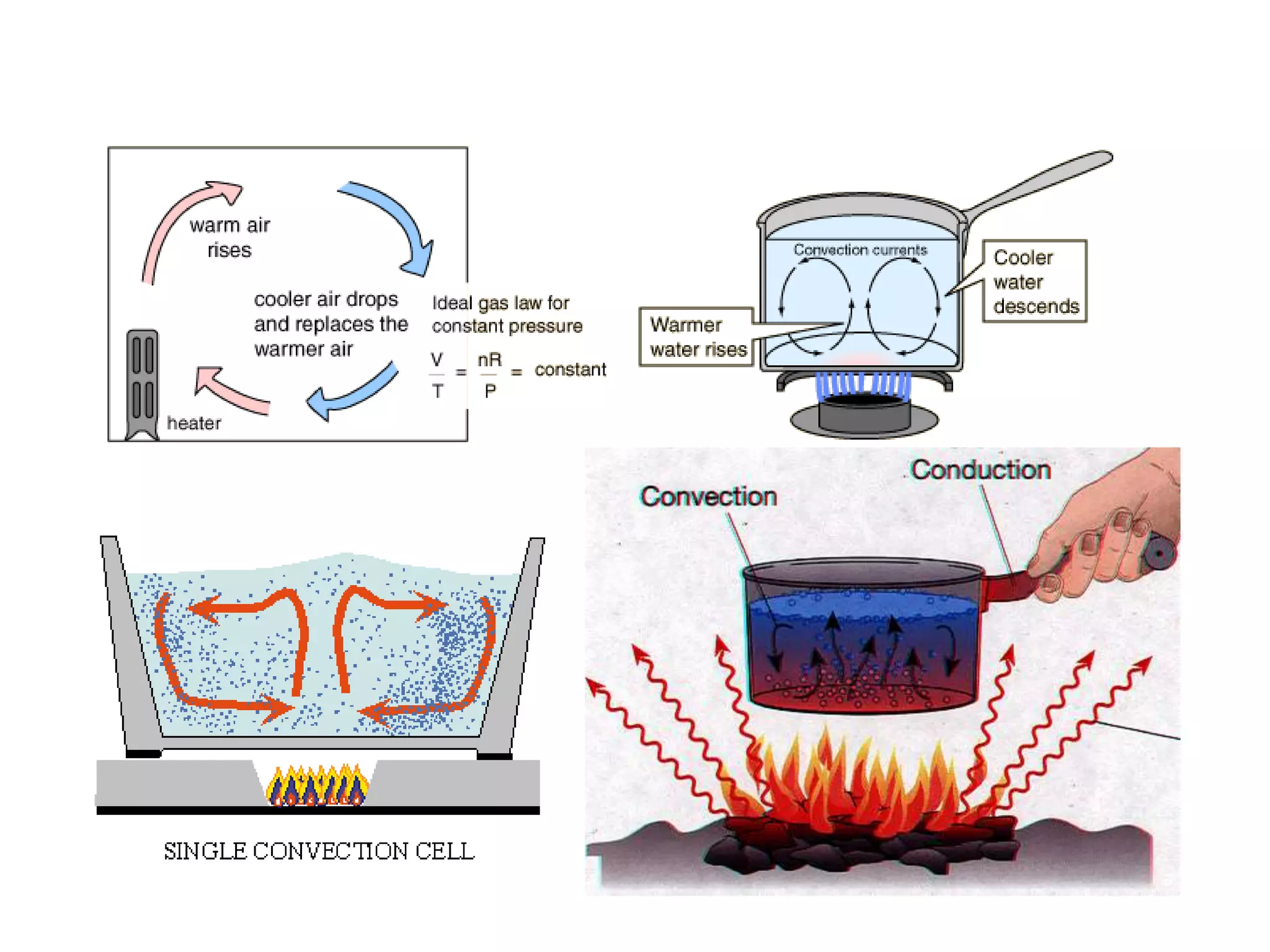
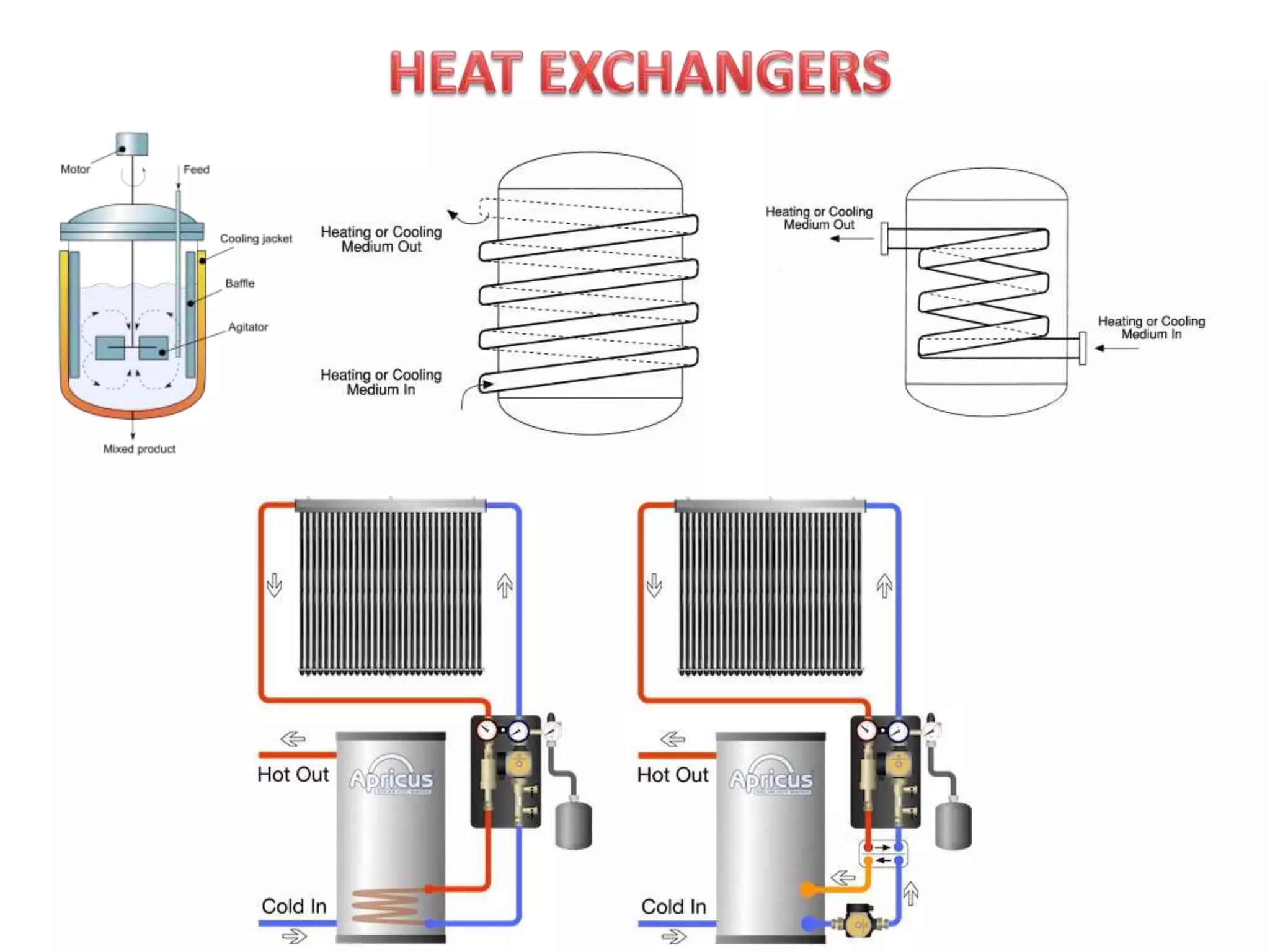
Heat is a form of energy that transfers from warmer objects to cooler ones. There are three main modes of heat transfer: conduction, convection, and radiation. Conduction involves the transfer of kinetic energy between adjacent particles in a substance. Convection refers to the movement of heated parts of fluids like gases and liquids. Radiation involves the emission and absorption of electromagnetic waves between objects and does not require a medium. Heat transfer aims to equalize temperatures and is driven by temperature differences between systems.