Pyridine- Pharmacy-Heterocyclic chemistry
•
15 likes•1,029 views
Pyridine- Pharmacy-Heterocyclic chemistry- As per PCI syllabus
Report
Share
Report
Share
Recommended
Heterocyclic compounds pyrazole

Heterocyclic compounds pyrazoleMinal Saini , student of Chaudhary bansilal University , Bhiwani, Haryana
More Related Content
What's hot
Heterocyclic compounds pyrazole

Heterocyclic compounds pyrazoleMinal Saini , student of Chaudhary bansilal University , Bhiwani, Haryana
What's hot (20)
Oxazole - Synthesis of Oxazole - Reactions of Oxazole - Medicinal uses of Oxa...

Oxazole - Synthesis of Oxazole - Reactions of Oxazole - Medicinal uses of Oxa...
Heterocyclic compounds - Furan - Synthesis of furan - Characteristic reaction...

Heterocyclic compounds - Furan - Synthesis of furan - Characteristic reaction...
Synthesis, reactivity, aromatic character and importance of Pyridine

Synthesis, reactivity, aromatic character and importance of Pyridine
Similar to Pyridine- Pharmacy-Heterocyclic chemistry
Similar to Pyridine- Pharmacy-Heterocyclic chemistry (19)
CHEMISTRY AND SYNTHESIS OF PYRIMIDINE - HETEROCYCLIC CHEMISTRY

CHEMISTRY AND SYNTHESIS OF PYRIMIDINE - HETEROCYCLIC CHEMISTRY
Heterocyclic Compounds Part-IV (Pyridine) by Dr Pramod R Padole

Heterocyclic Compounds Part-IV (Pyridine) by Dr Pramod R Padole
Heterocyclic compounds - pyrrole - synthesis of pyrrole - characteristic rea...

Heterocyclic compounds - pyrrole - synthesis of pyrrole - characteristic rea...
Porphyrins introduction applications synthesis.pptx

Porphyrins introduction applications synthesis.pptx
Heterocyclic Compounds Part-II (Pyridine) by Dr Pramod R Padole

Heterocyclic Compounds Part-II (Pyridine) by Dr Pramod R Padole
More from Akhil Nagar
More from Akhil Nagar (20)
Cephalosporins antibiotics - Beta lactam antibiotics

Cephalosporins antibiotics - Beta lactam antibiotics
Sulphonamides- Anti bacterial- Medicinal Chemistry-Pharmacy

Sulphonamides- Anti bacterial- Medicinal Chemistry-Pharmacy
Anthelmintic drugs- Treatment - Pharmacy- Medicinal Chemistry

Anthelmintic drugs- Treatment - Pharmacy- Medicinal Chemistry
Urinary Tract Infection and Treatment-Pharmacy- Medicinal Chemistry lecture n...

Urinary Tract Infection and Treatment-Pharmacy- Medicinal Chemistry lecture n...
Malaria and Antimalarial Drugs and recent Advancement 

Malaria and Antimalarial Drugs and recent Advancement
Dakin reaction- organic and Heterocyclic chemistry- As per PCI syllabus

Dakin reaction- organic and Heterocyclic chemistry- As per PCI syllabus
Beckmann rearrangement- Organic and Heterocyclic chemistry- As per PCI syllabus

Beckmann rearrangement- Organic and Heterocyclic chemistry- As per PCI syllabus
Wolff kishner reduction, Organic and heterocyclic chemistry, As per PCI sylll...

Wolff kishner reduction, Organic and heterocyclic chemistry, As per PCI sylll...
Clemmensen reduction- Heterocyclic and Organic chemistry- As per PCI syllabus

Clemmensen reduction- Heterocyclic and Organic chemistry- As per PCI syllabus
Recently uploaded
Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝9953056974 Low Rate Call Girls In Saket, Delhi NCR
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptxVS Mahajan Coaching Centre
“Oh GOSH! Reflecting on Hackteria's Collaborative Practices in a Global Do-It...

“Oh GOSH! Reflecting on Hackteria's Collaborative Practices in a Global Do-It...Marc Dusseiller Dusjagr
Recently uploaded (20)
Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝

Model Call Girl in Bikash Puri Delhi reach out to us at 🔝9953056974🔝
Organic Name Reactions for the students and aspirants of Chemistry12th.pptx

Organic Name Reactions for the students and aspirants of Chemistry12th.pptx
Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...

Kisan Call Centre - To harness potential of ICT in Agriculture by answer farm...
A Critique of the Proposed National Education Policy Reform

A Critique of the Proposed National Education Policy Reform
“Oh GOSH! Reflecting on Hackteria's Collaborative Practices in a Global Do-It...

“Oh GOSH! Reflecting on Hackteria's Collaborative Practices in a Global Do-It...
Contemporary philippine arts from the regions_PPT_Module_12 [Autosaved] (1).pptx![Contemporary philippine arts from the regions_PPT_Module_12 [Autosaved] (1).pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
![Contemporary philippine arts from the regions_PPT_Module_12 [Autosaved] (1).pptx](data:image/gif;base64,R0lGODlhAQABAIAAAAAAAP///yH5BAEAAAAALAAAAAABAAEAAAIBRAA7)
Contemporary philippine arts from the regions_PPT_Module_12 [Autosaved] (1).pptx
Incoming and Outgoing Shipments in 1 STEP Using Odoo 17

Incoming and Outgoing Shipments in 1 STEP Using Odoo 17
Pyridine- Pharmacy-Heterocyclic chemistry
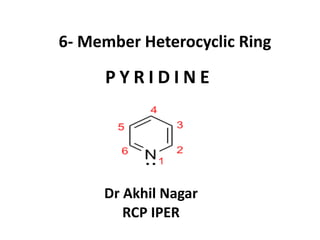
- 1. 6- Member Heterocyclic Ring P Y R I D I N E Dr Akhil Nagar RCP IPER
- 2. Properties 1. Aromaticity P Y R I D I N E
- 4. Properties 2. Basicity P Y R I D I N E
- 5. Properties 2. Basicity Pyridine is more basic than pyrrole P Y R I D I N E 113
- 9. 123
- 10. Reactions 124
- 11. Resonating Structures: The bond length and stability of Pyridine can be explained by the resonating structures. Electronegative atom N produces the deficiency of electron density in pyridine and ring carbon acquires increased electron density. Thus nitrogen atom in pyridine causes deactivation of the ring, contrary to pyrrole.
- 12. 2018 125 C2 C3 C4 Reactions 2. Electrophilic substitution to C
- 15. 128 nucleophilic substitution Reactions 3. Nucleophilic substitution Why pyridine undergoes reaction at 2-position. C3 C2
- 16. Reactions Alkylation of Pyridine gives 2-butylpyridine
- 17. Reactions Reaction with Potassium Hydroxide
- 19. 130 Reactions 4. Pyridine as Nucleophilic catalyst
- 21. 117 Synthesis 1. Hantzsch pyridine synthesis
- 22. 118 Synthesis 1. Hantzsch pyridine synthesis
- 26. 119 Synthesis 1. Hantzsch pyridine synthesis
- 27. 120 Synthesis 2. The Guareschi Synthesis
- 28. 121 Synthesis 3. From 1,5 - Dicarbonyl Compounds
