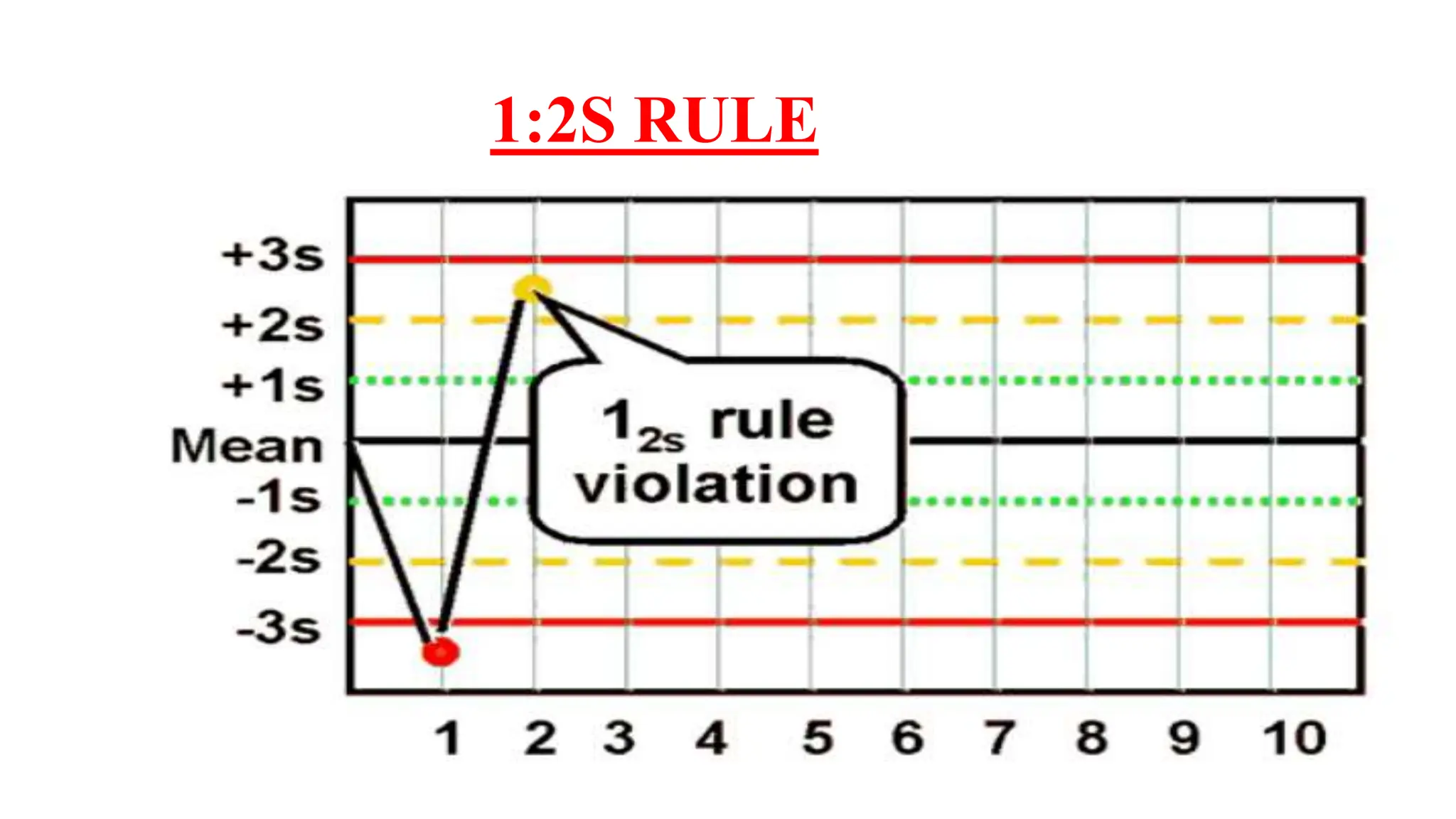
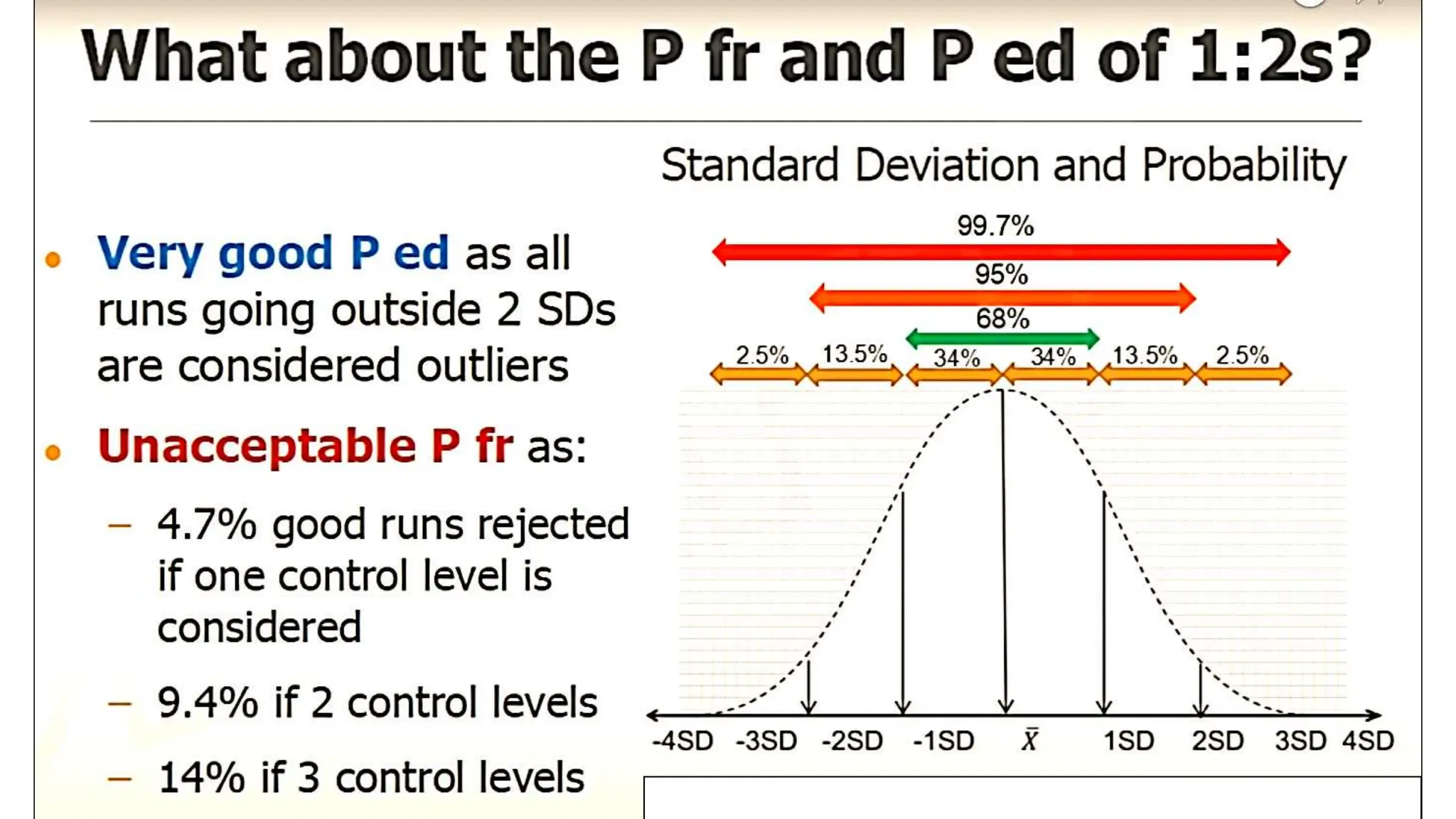
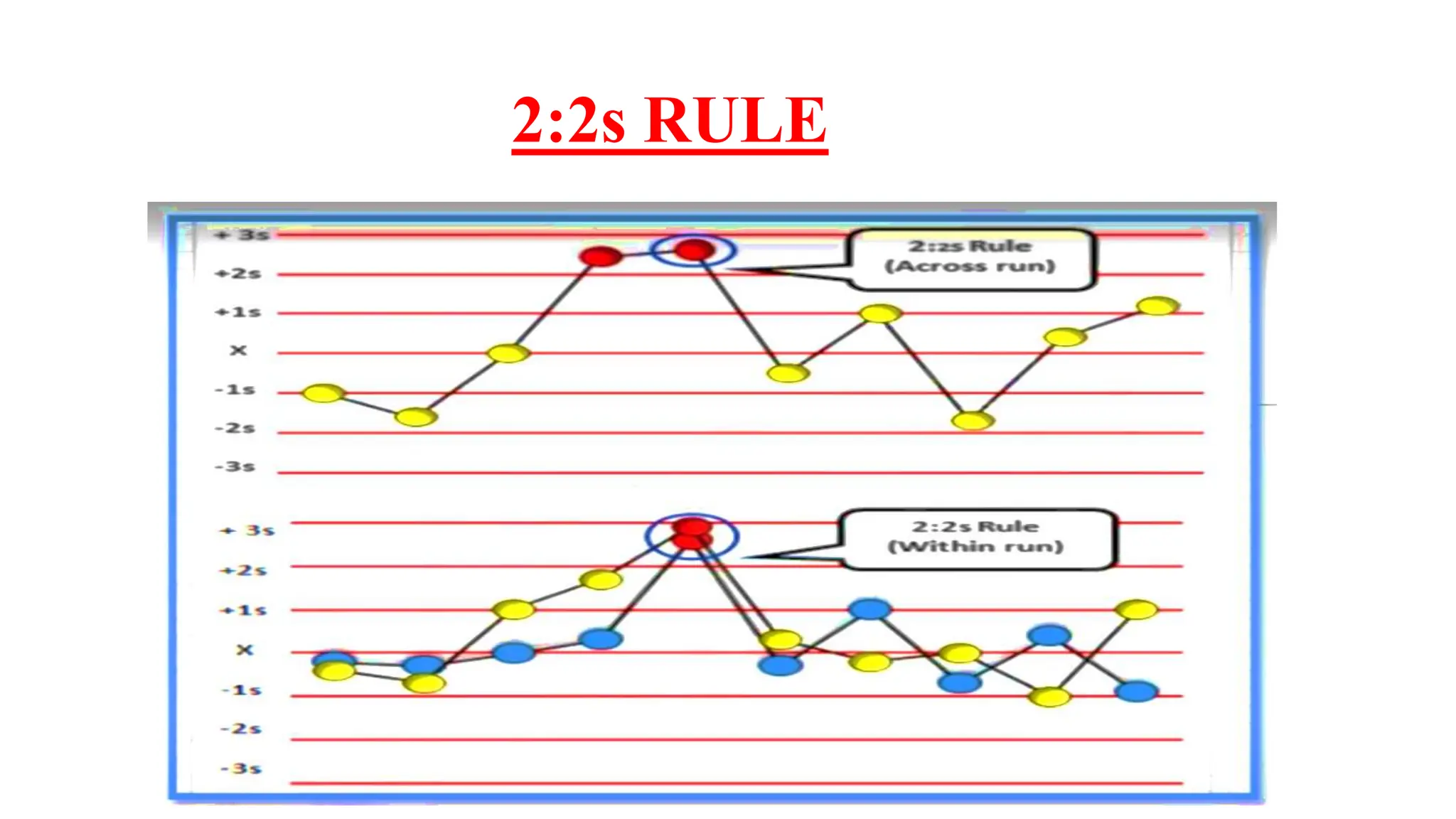
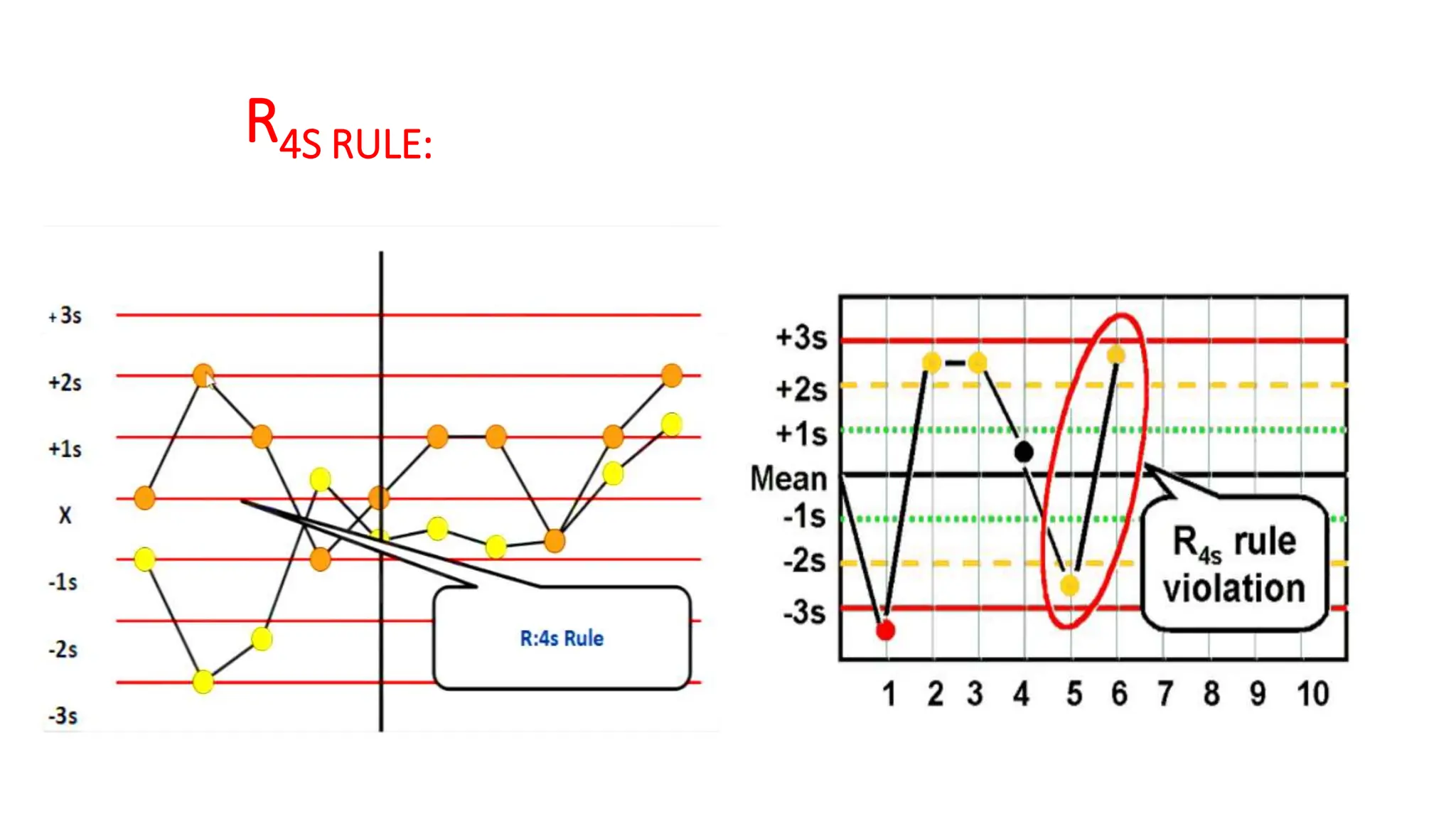
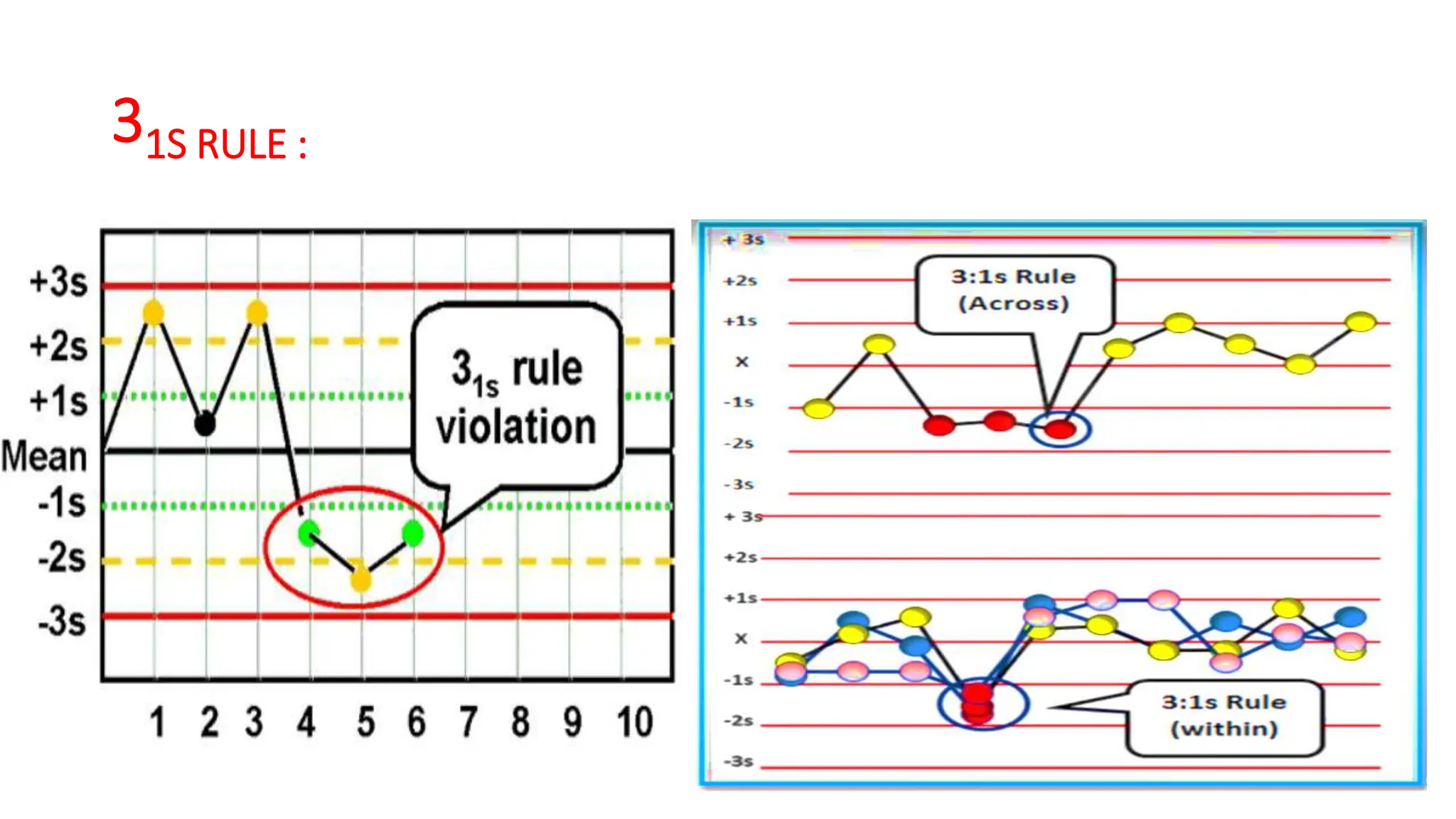
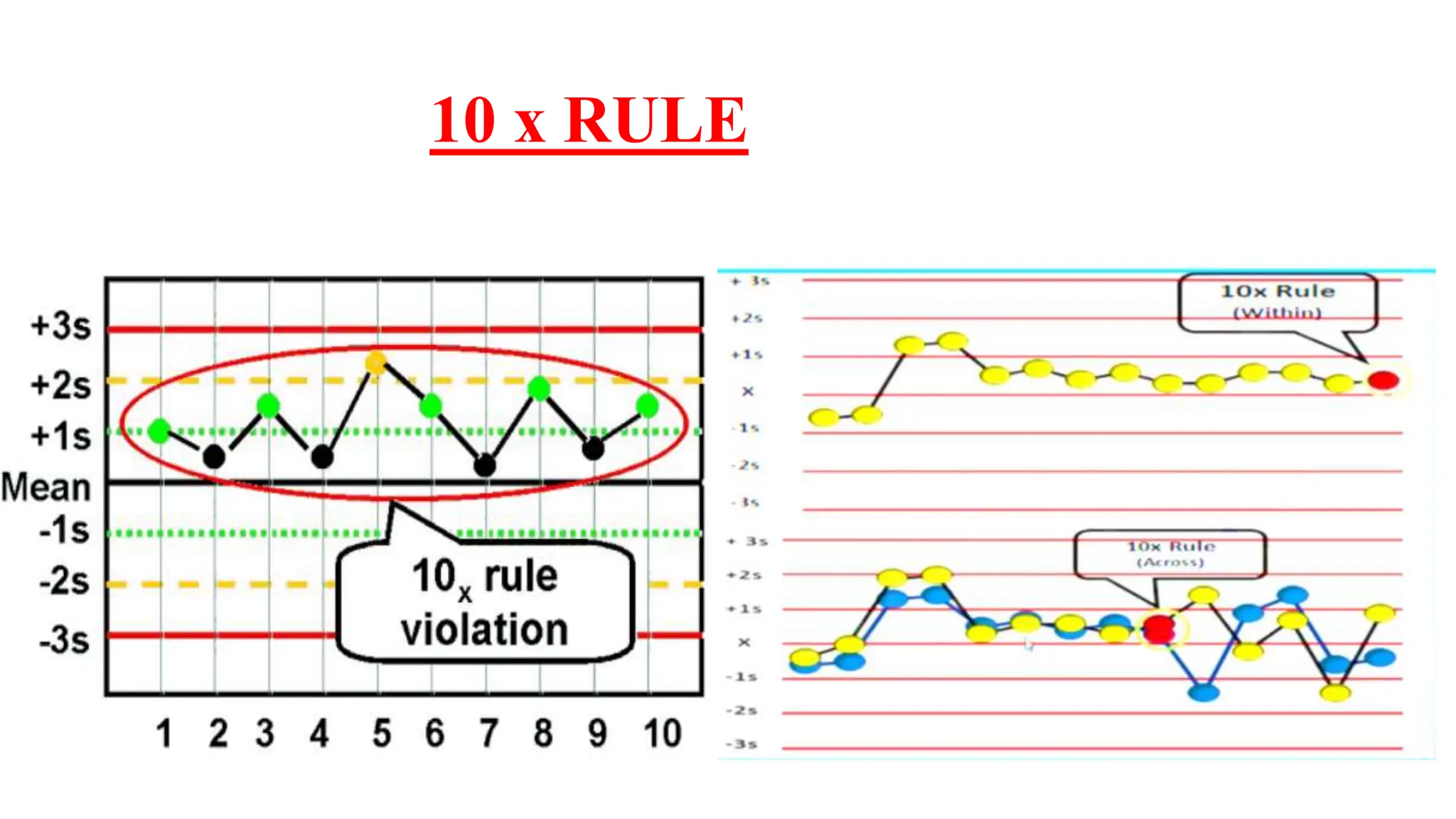
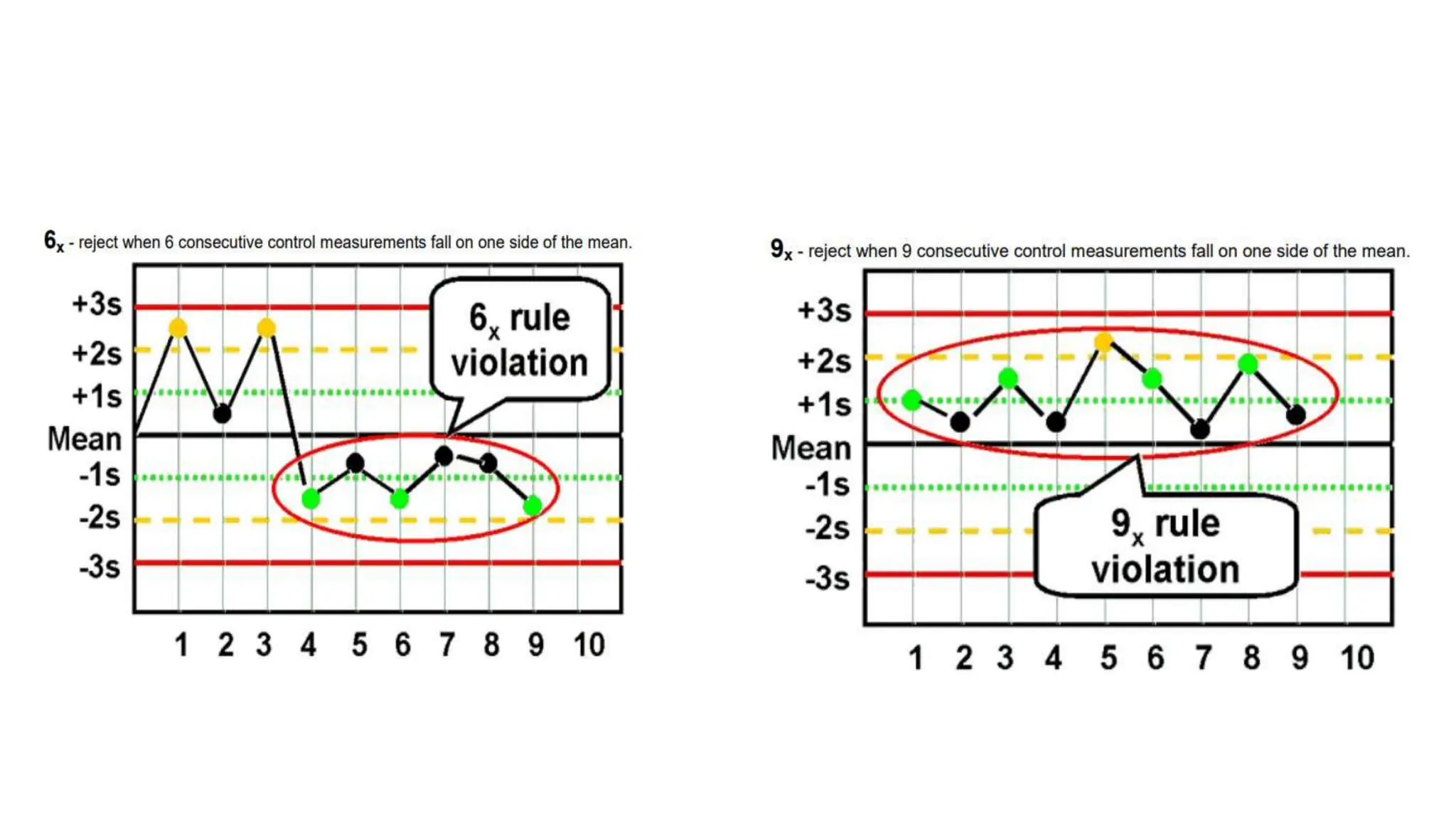
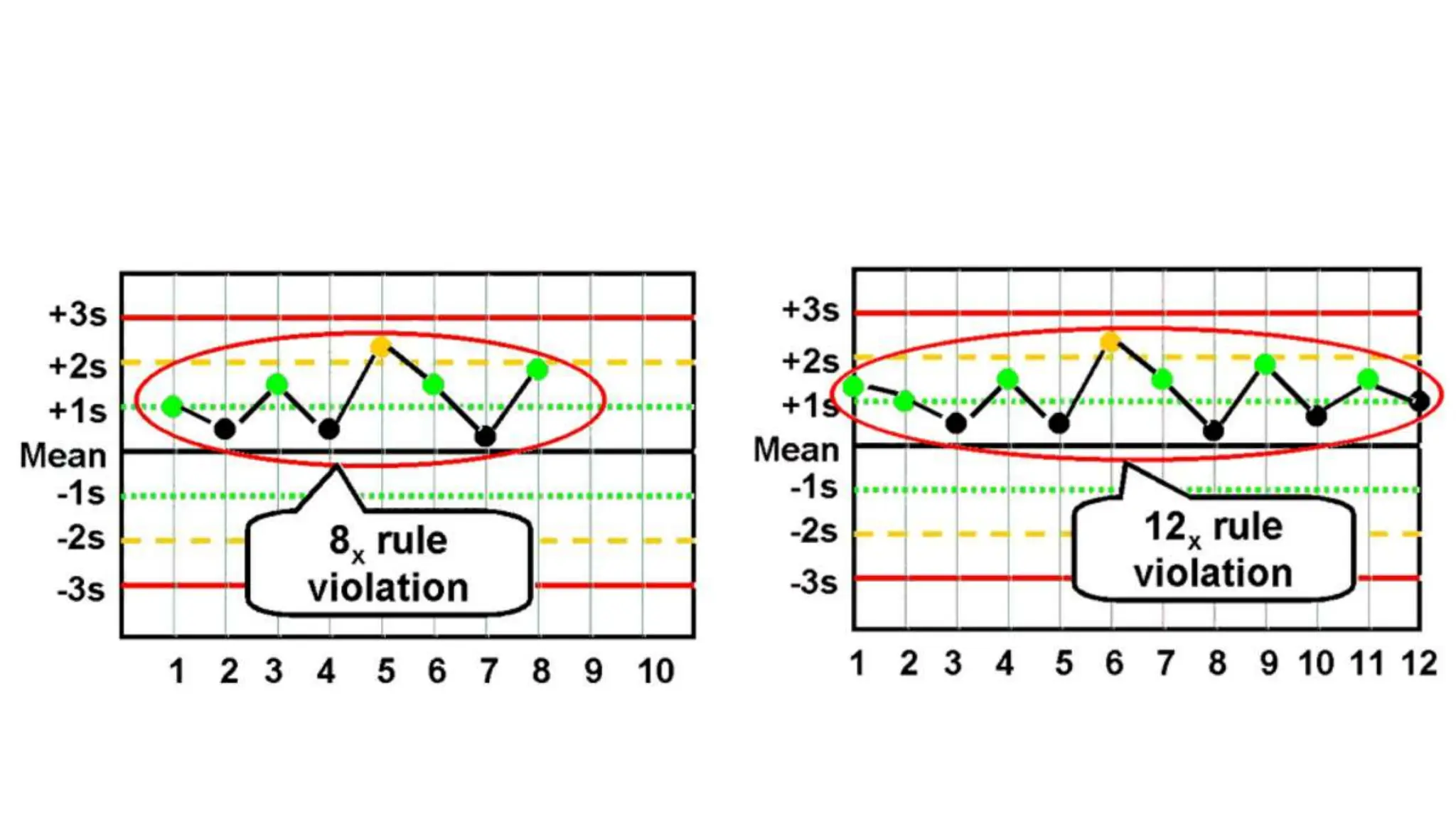
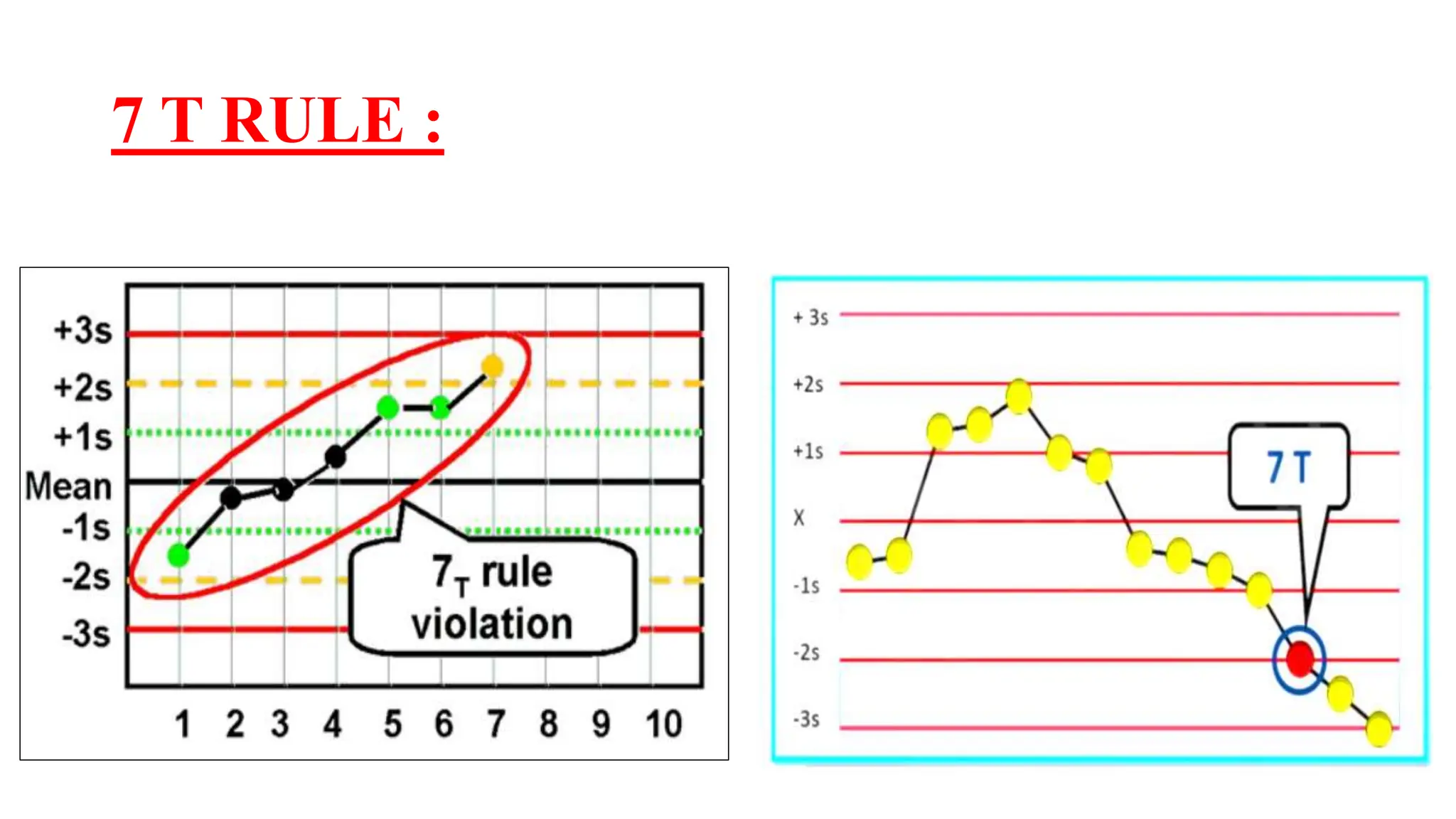
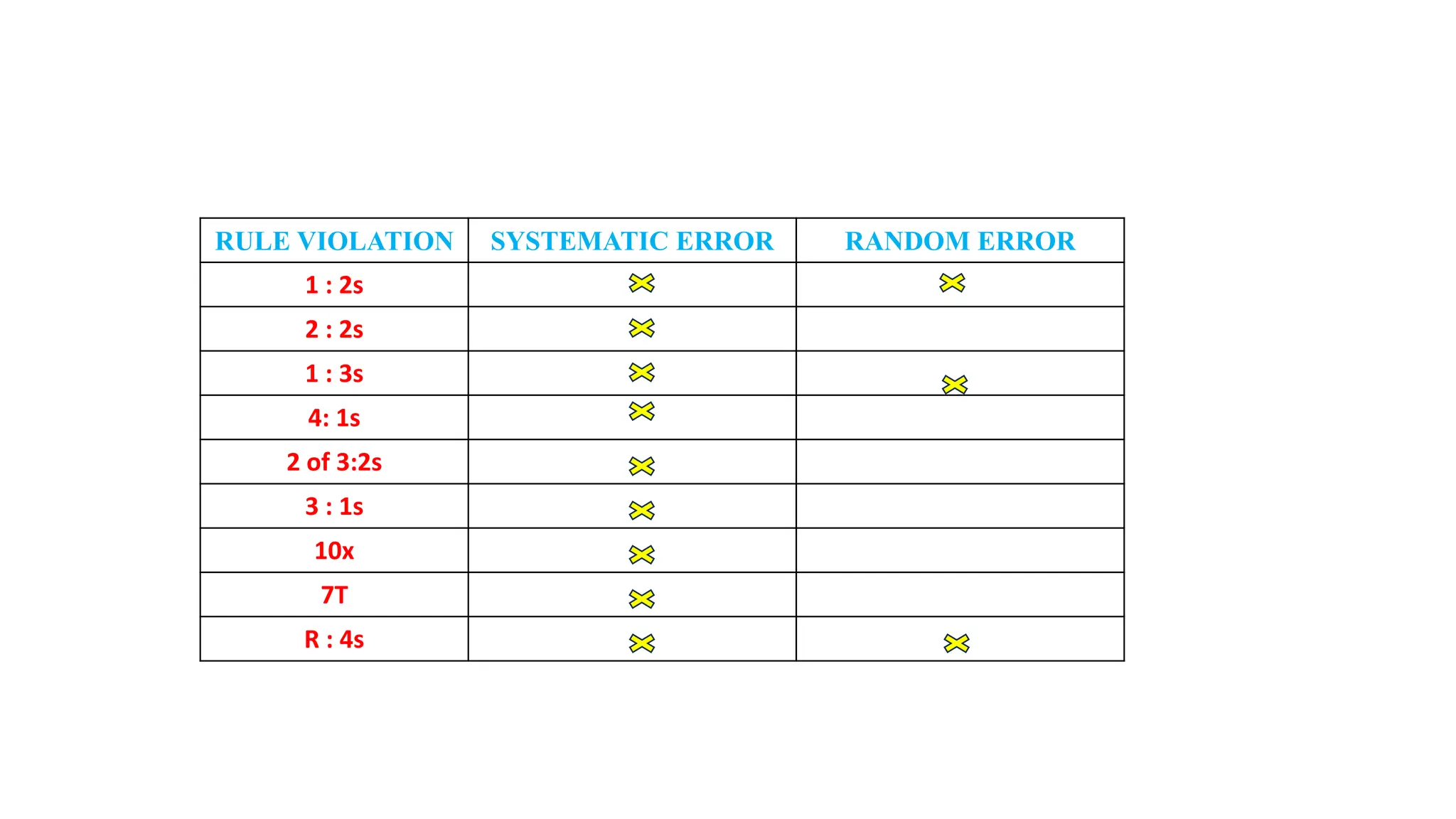
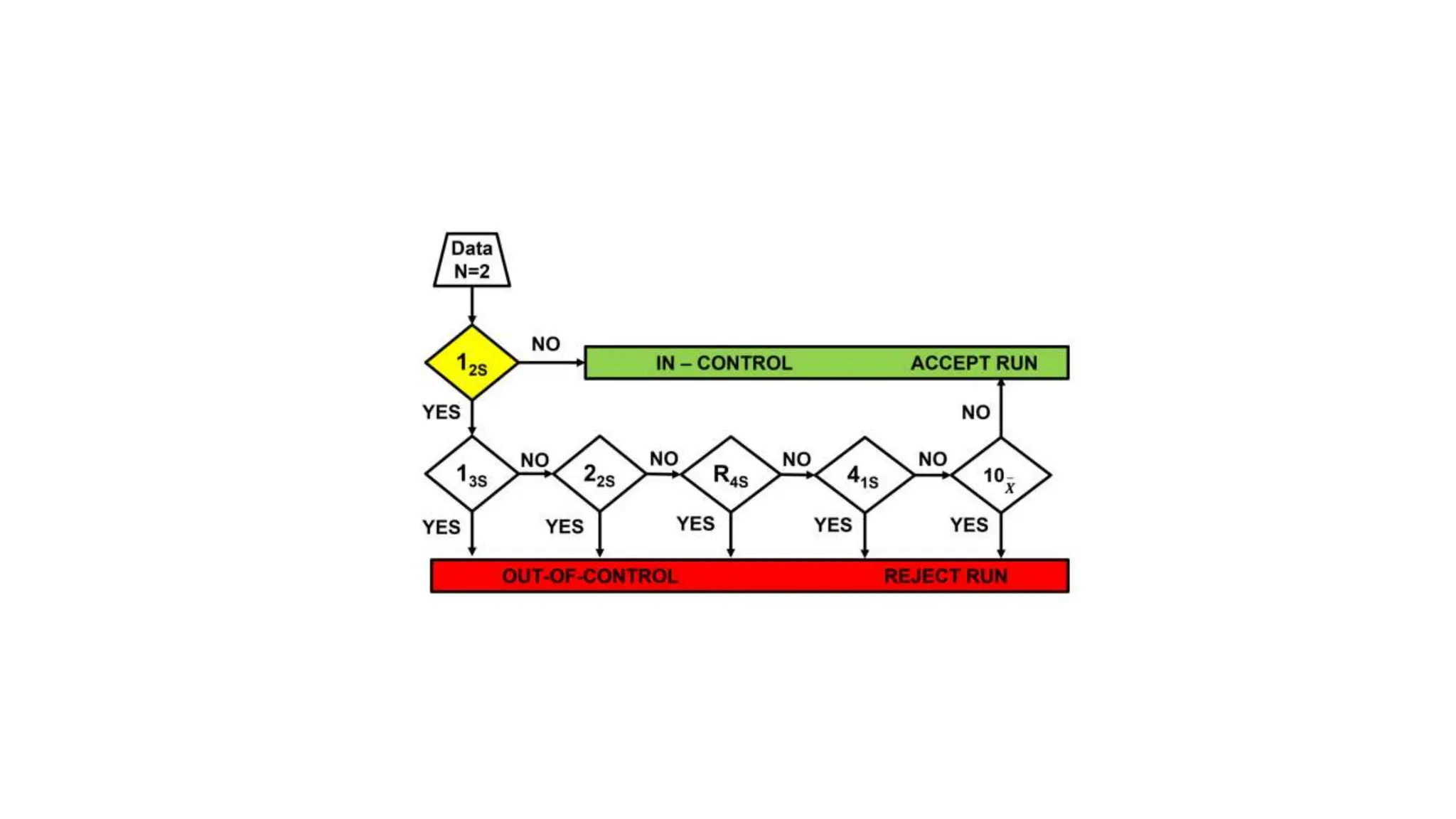
The Westgard rules provide a statistical framework for evaluating the quality of analytical runs in medical laboratories. Developed by Dr. James Westgard in the 1980s, the rules are based on statistical process control and can be used individually or together to detect random and systematic errors. Key rules include 1:2s for warning of potential errors, 1:3s and 2:2s for rejecting runs with errors, and 10x for identifying systematic biases over multiple runs. Laboratories should aim to maximize error detection while minimizing false rejections when applying these rules.