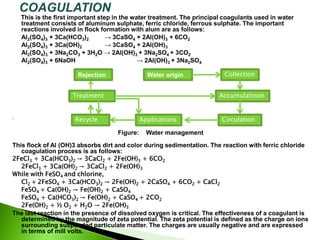
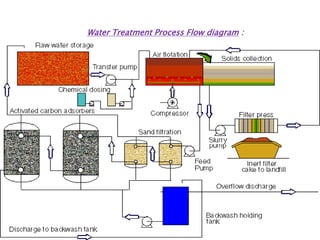
The document discusses the importance of water treatment in various industries, highlighting the necessity of removing impurities and regulating hardness for specific applications such as textile and food processing. It outlines different types of water sources (surface, subsoil, and deep well), describes key treatment processes (coagulation, filtration, disinfection, desalination), and explains methods for softening water to eliminate hardness caused by calcium and magnesium salts. Additionally, the document details chemical reactions involved in the treatment processes and emphasizes the challenges posed by impurities and hardness on industrial operations.