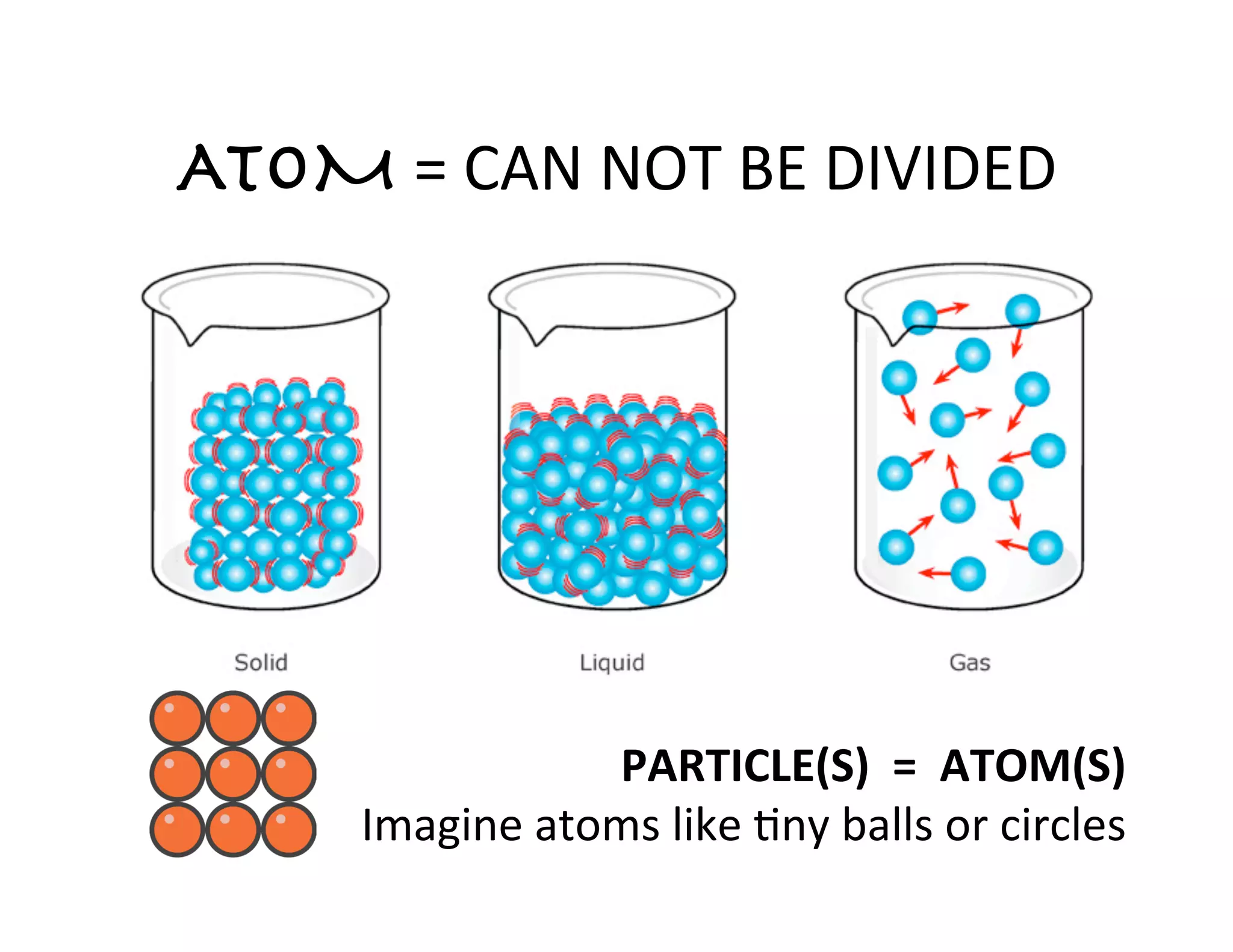
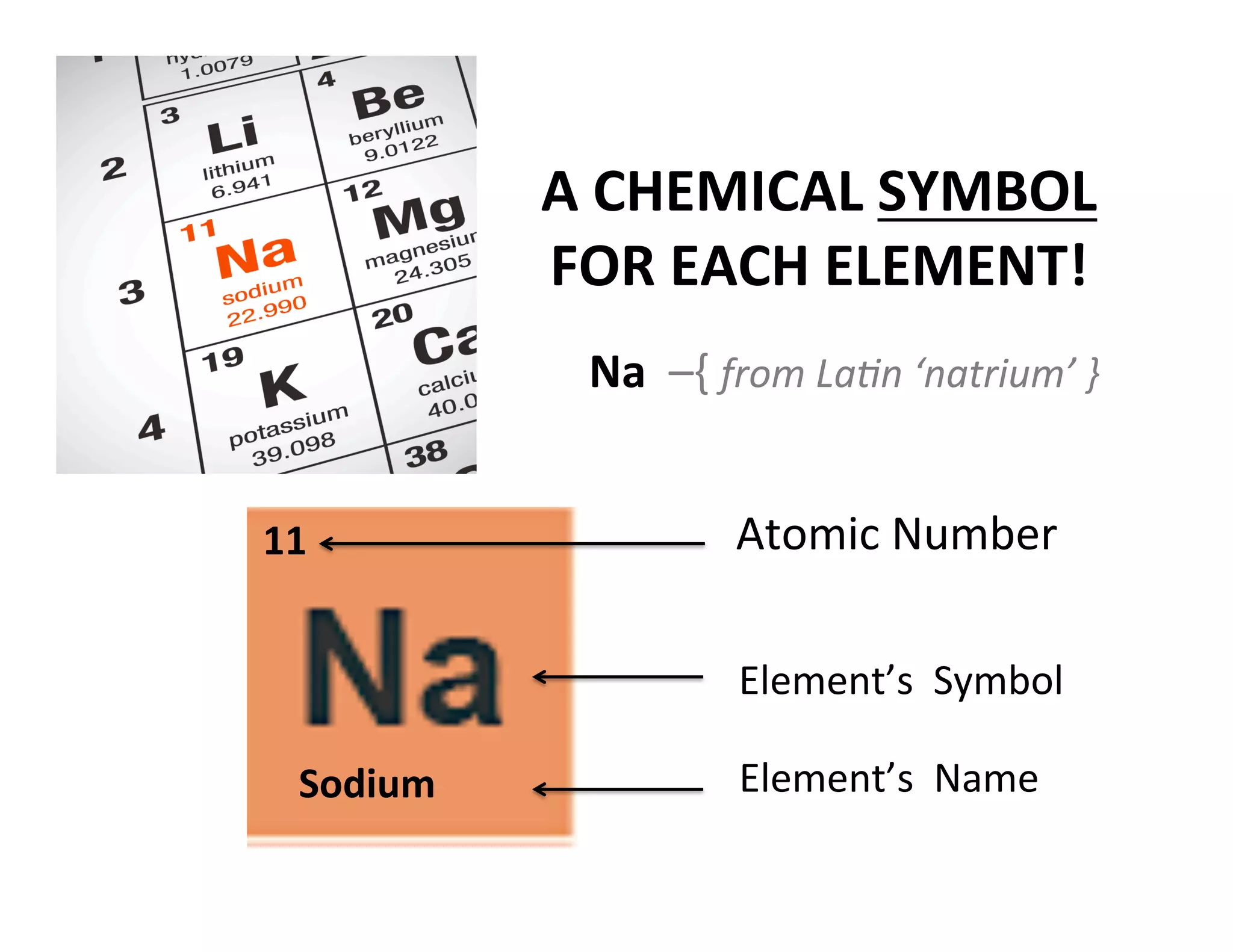
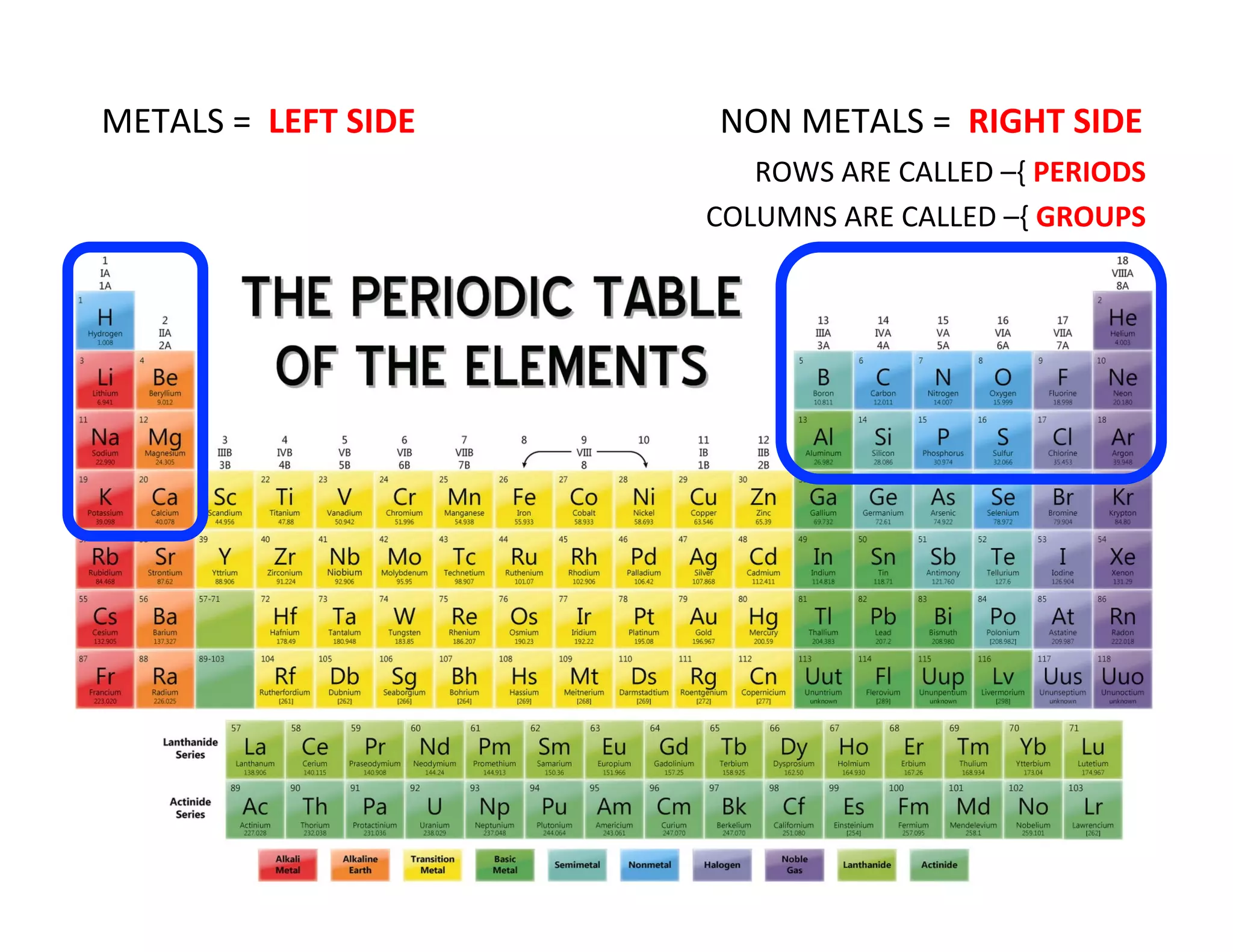
This document provides an overview of atomic theory and the elements. It defines atoms as indestructible particles that make up all matter and can differ in size, shape, mass and arrangement. Atoms of the same type form elements, which are represented by symbols and cannot be broken down further. There are 118 known elements, with 94 found naturally. The periodic table arranges elements in rows and columns based on atomic number and properties. Common questions are provided to help students learn about atoms and the first 20 elements.