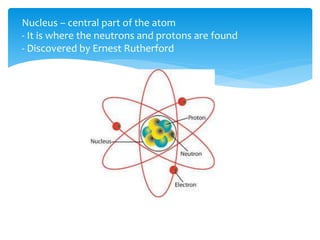
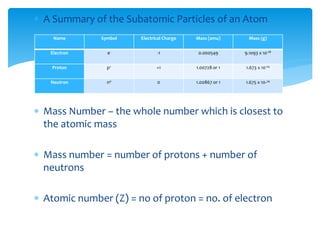
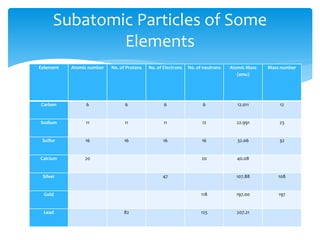
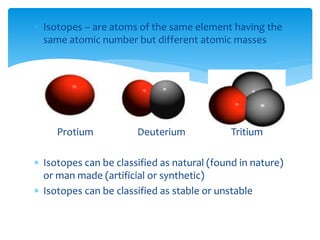
This document discusses the history and structure of the atom. It explains that ancient Greek philosophers first proposed that all matter is made of tiny indivisible particles called atoms. Over time, scientists discovered subatomic particles like electrons, protons, and neutrons that make up the nucleus of the atom. The document defines key terms like atomic number, mass number, isotopes, and provides examples of subatomic particles for some common elements.