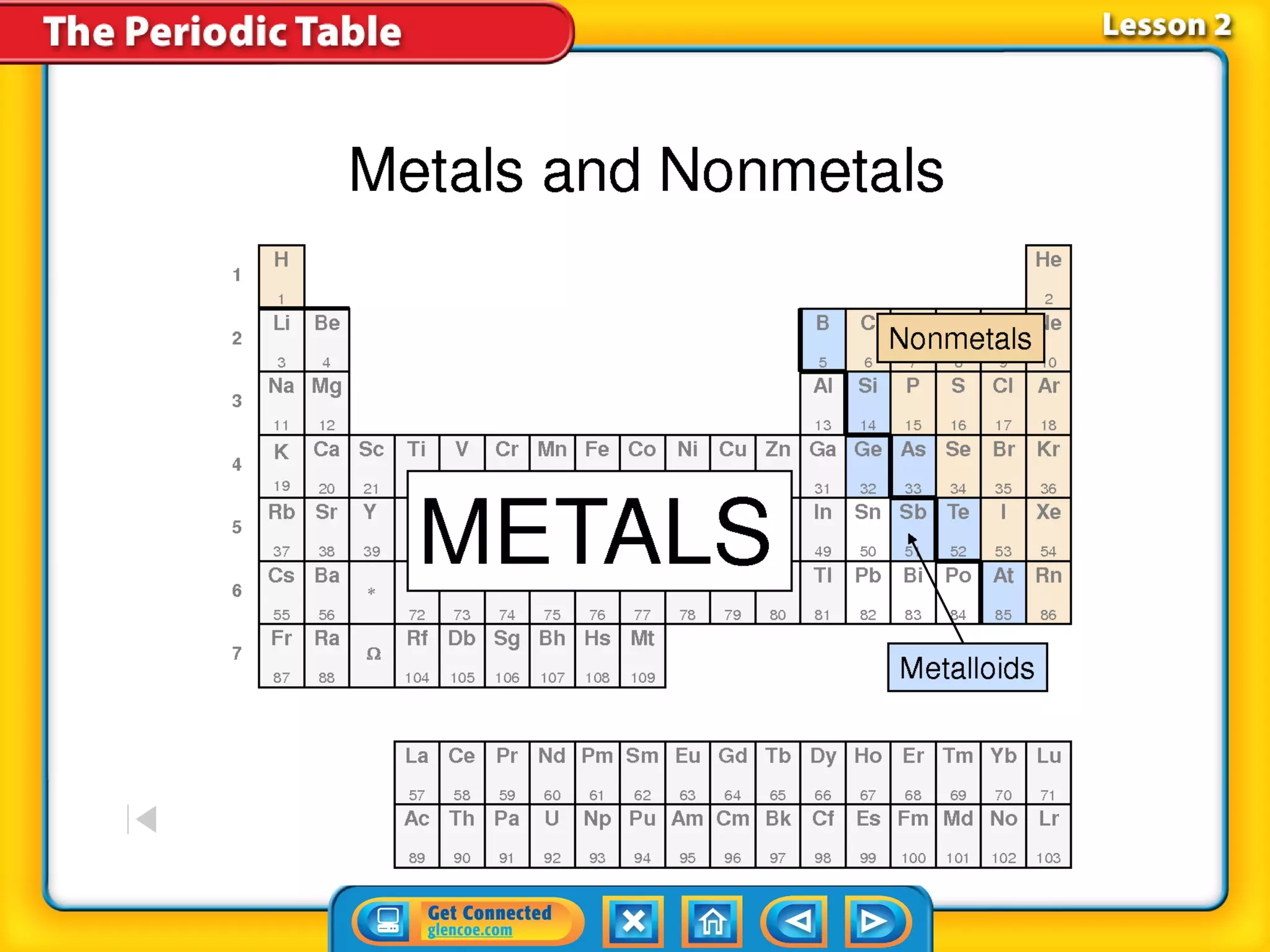
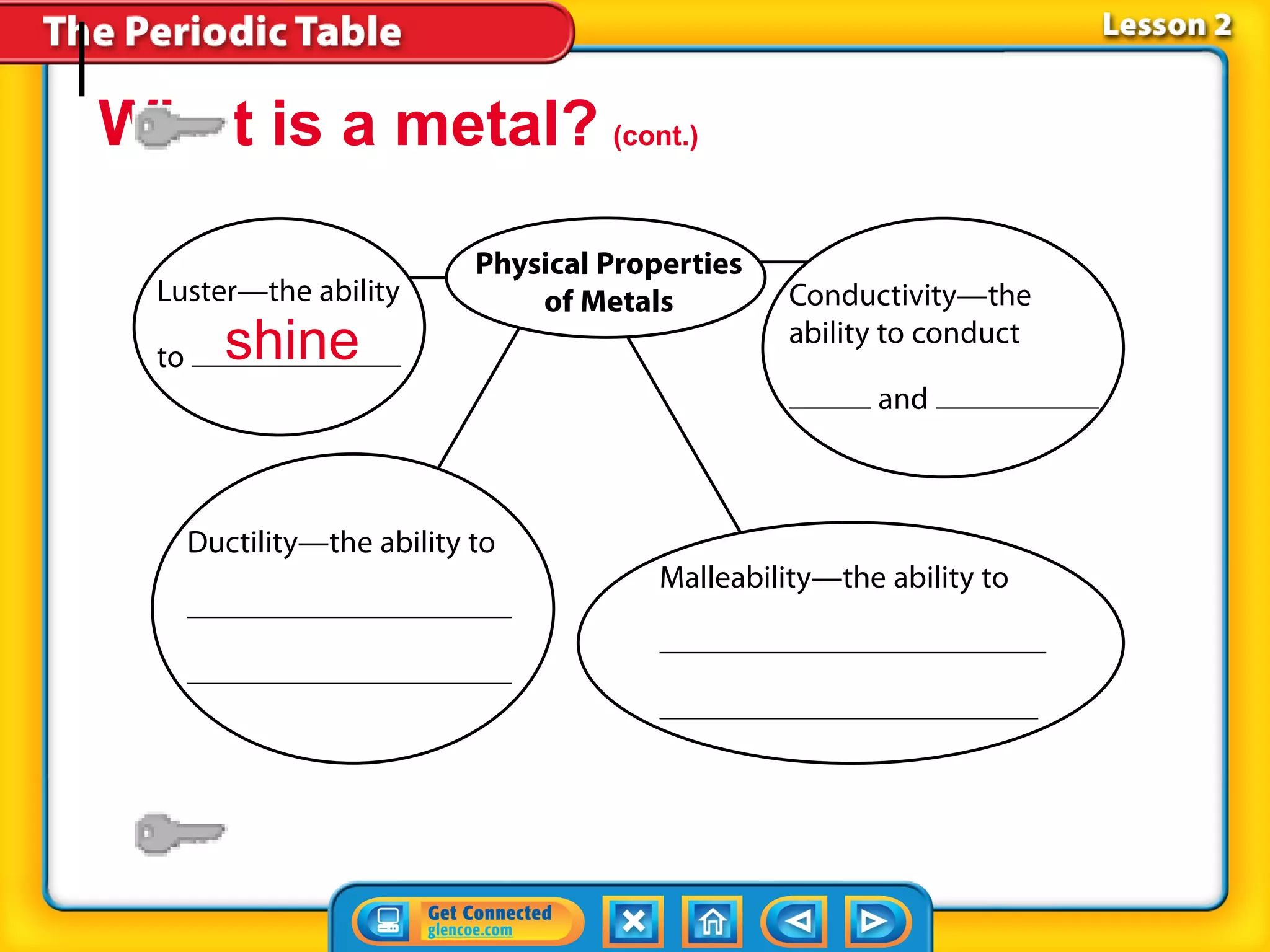
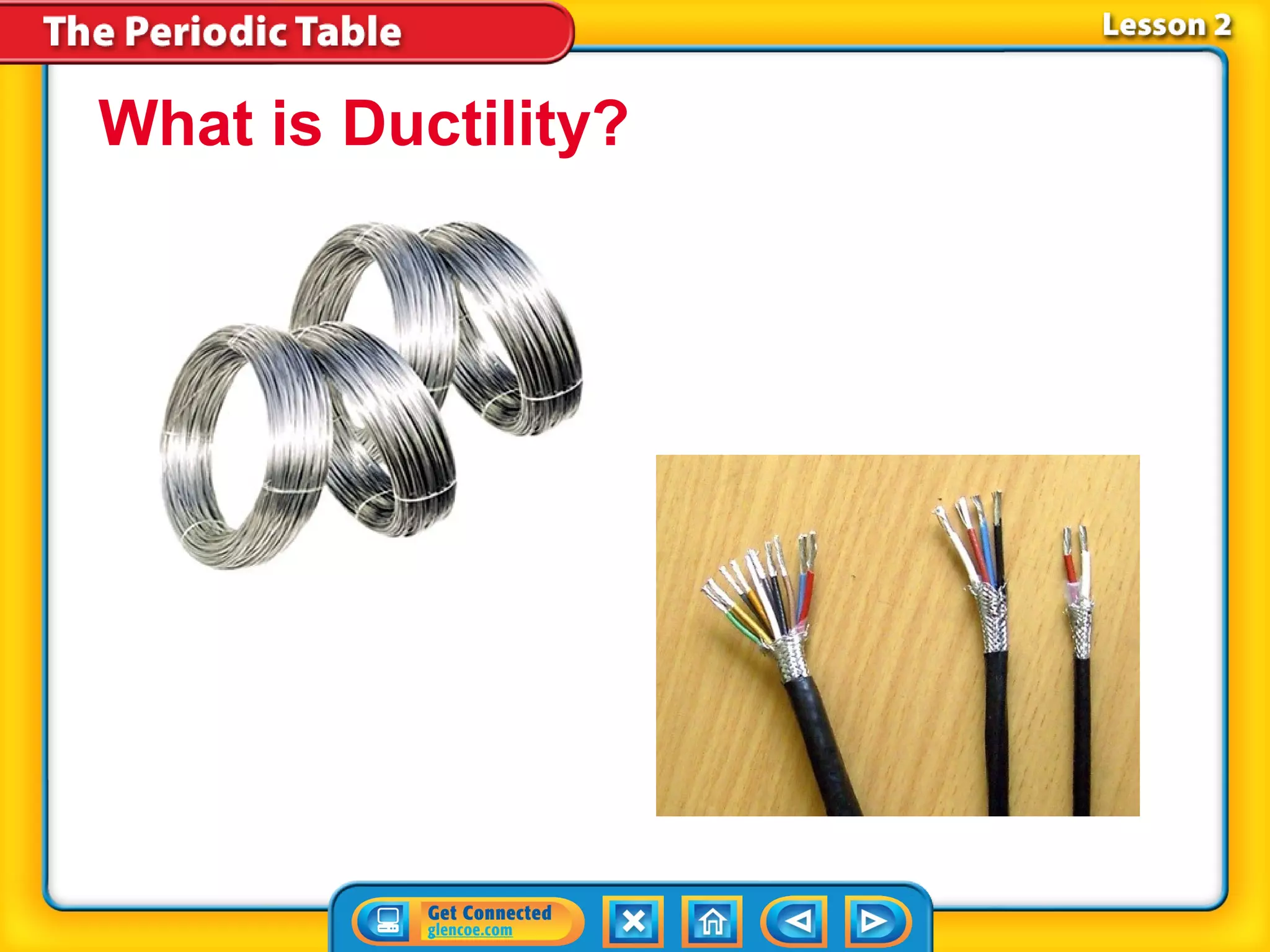
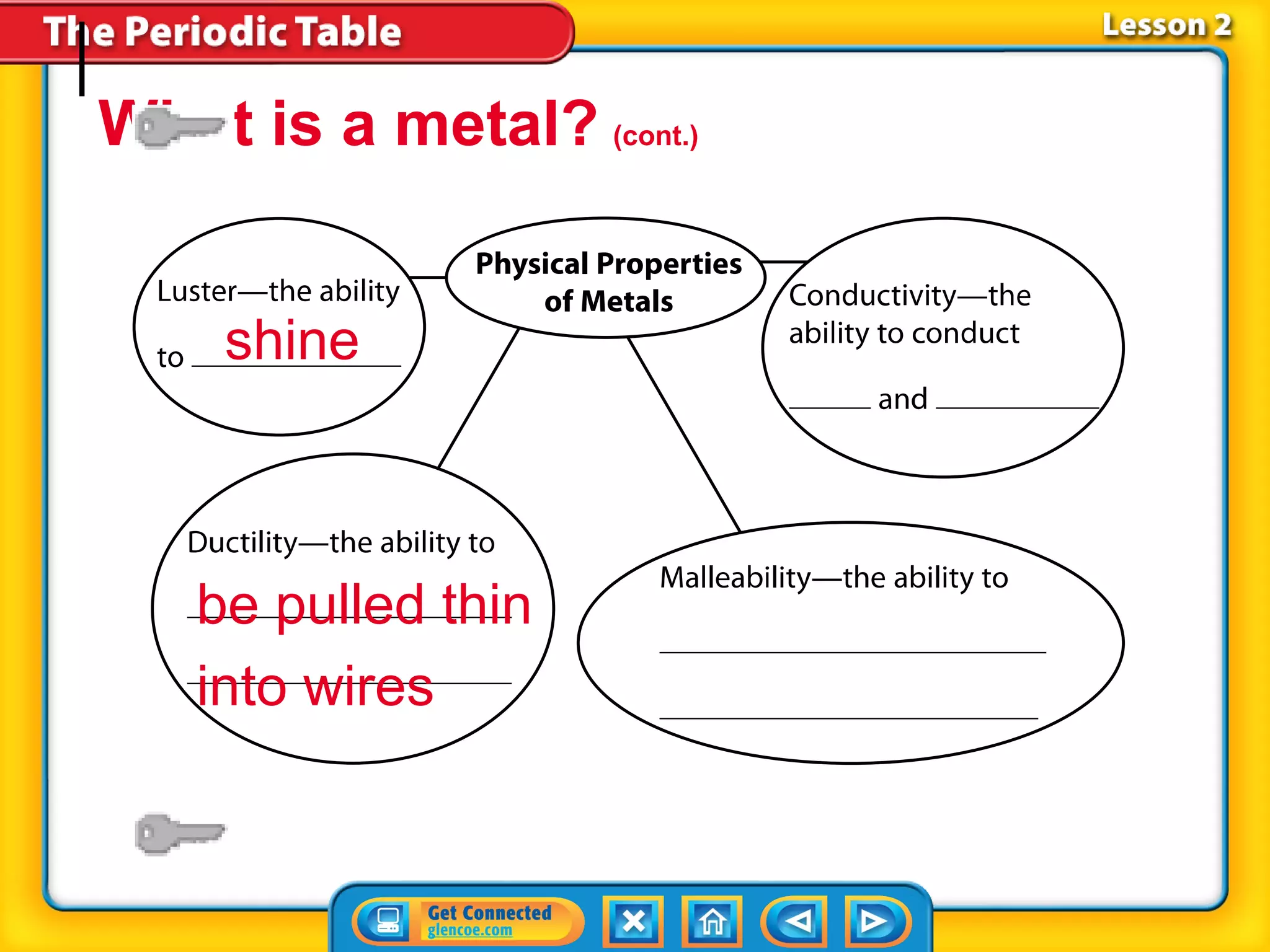
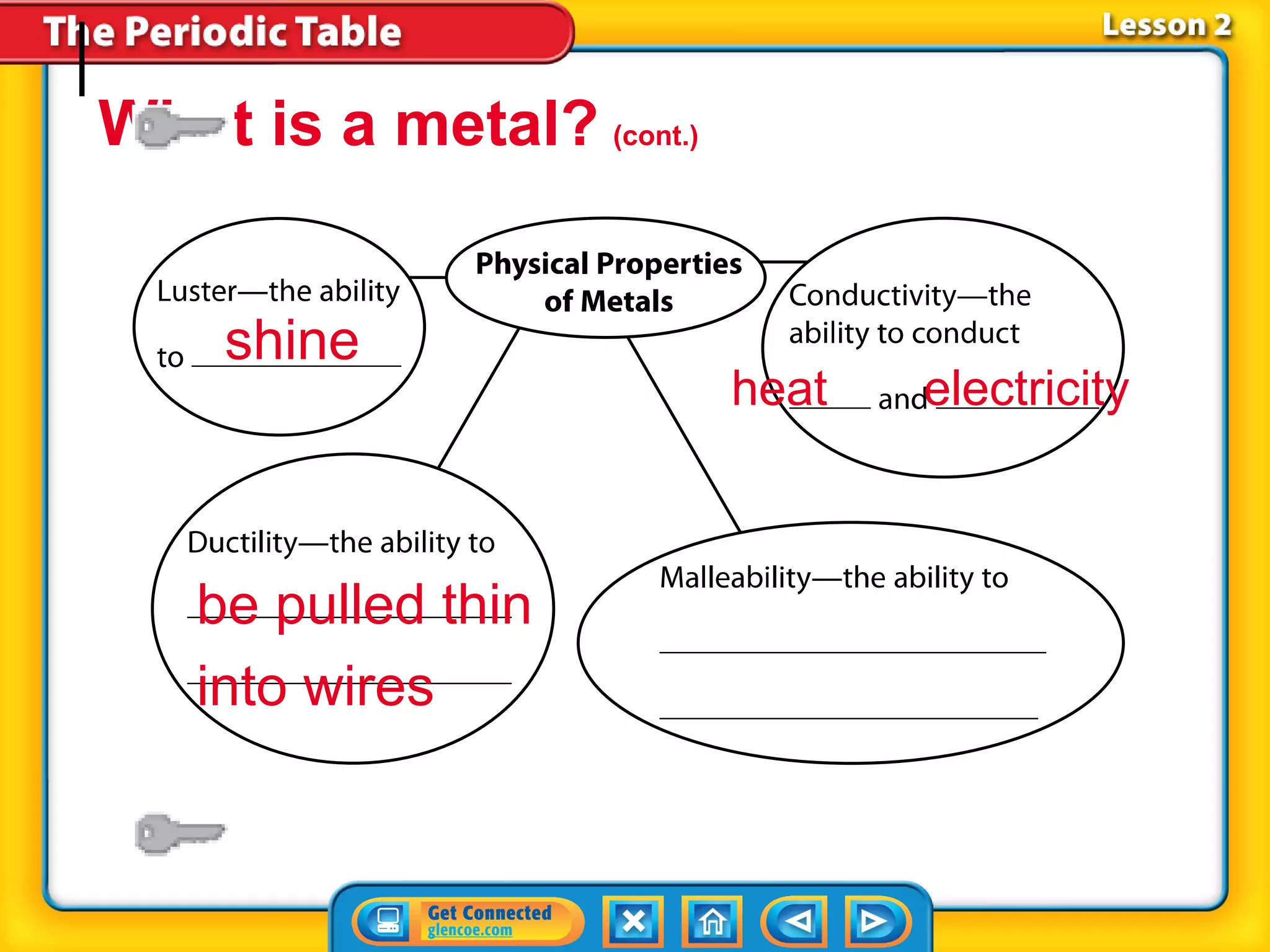
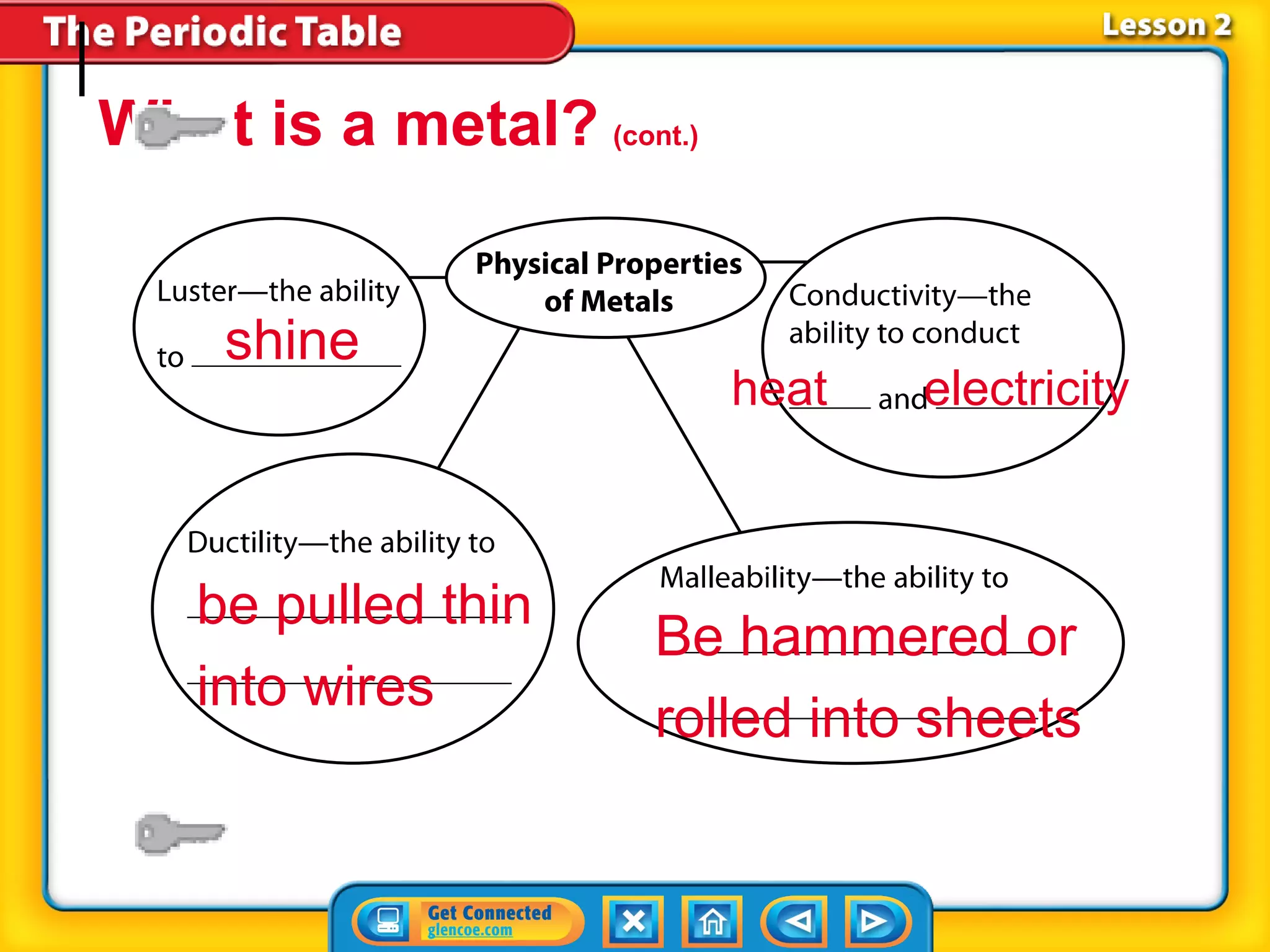
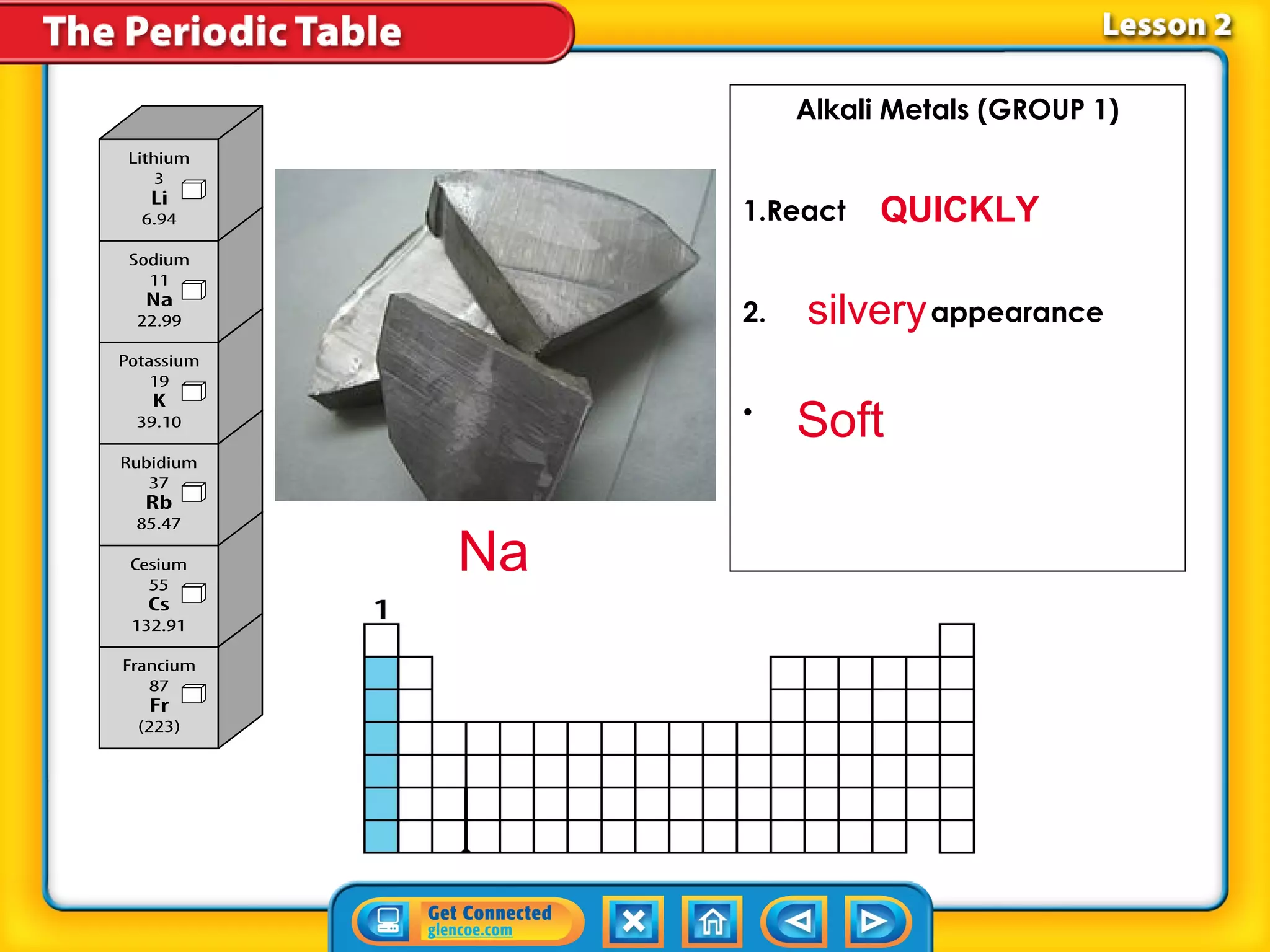
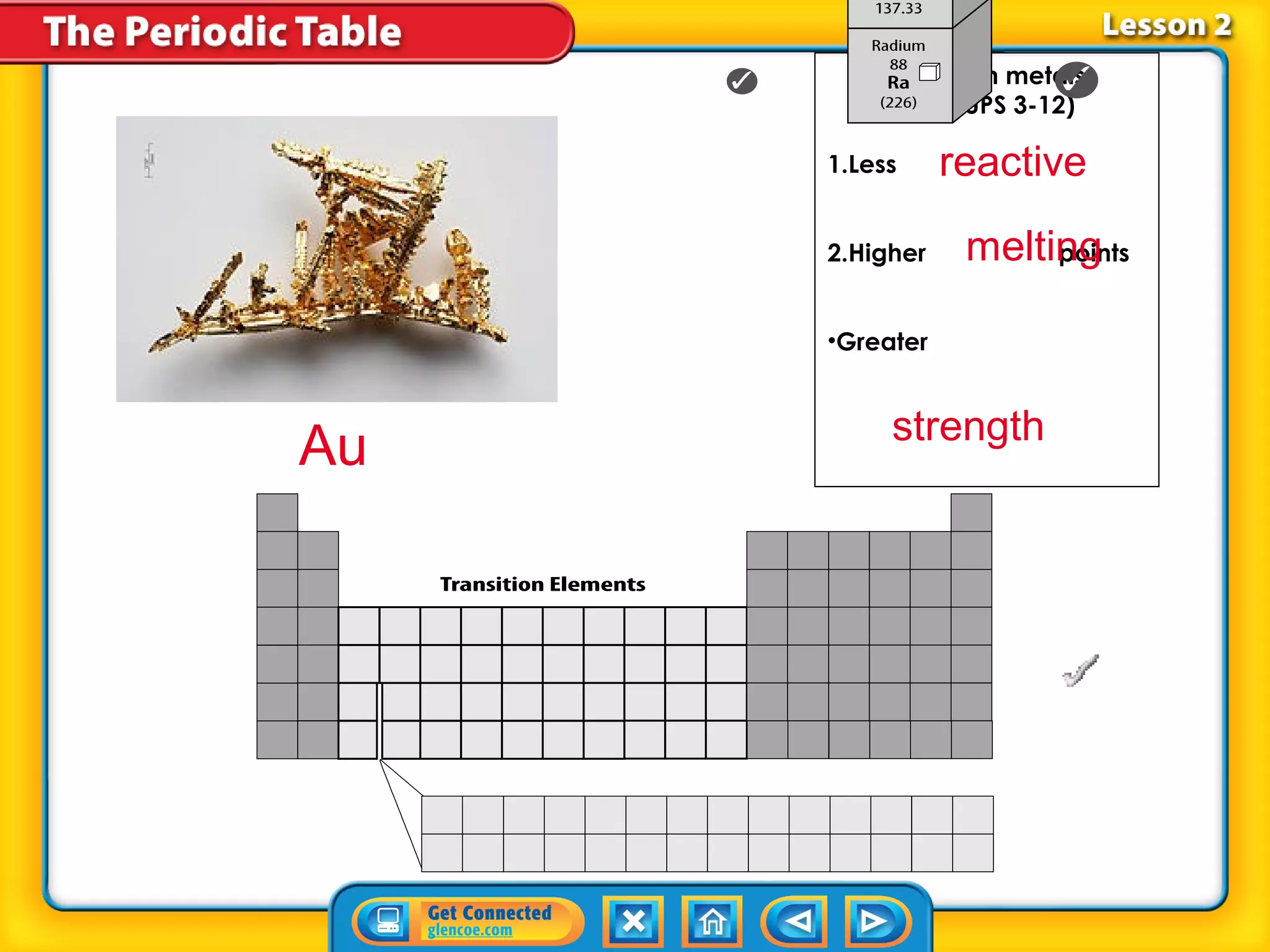
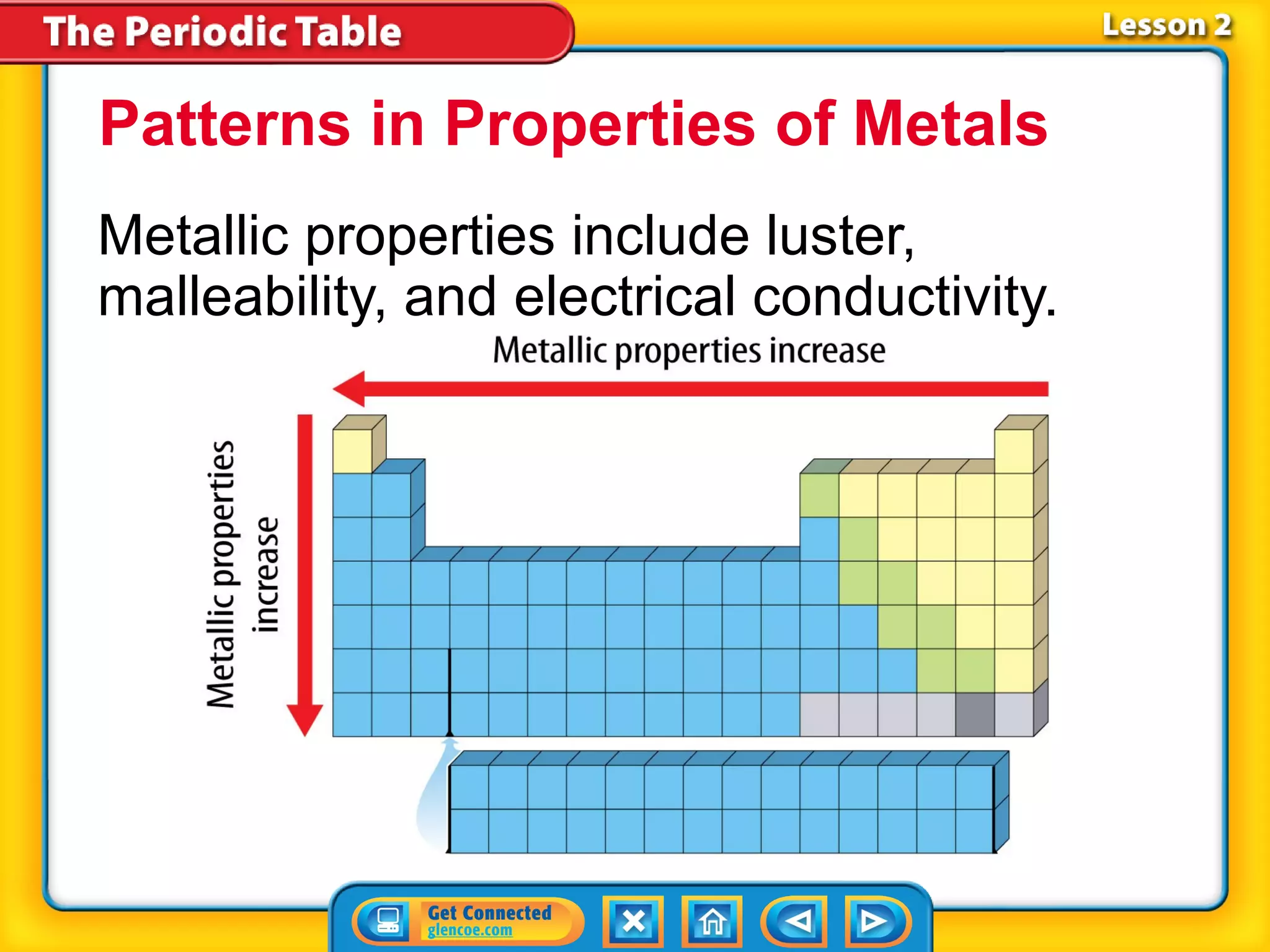
The document discusses the properties of metals. Metals are elements that are found in groups 1-2 and some in groups 13-15 on the periodic table. Key properties of metals include luster, ductility, malleability, and conductivity - metals are shiny, can be drawn into wires, hammered into thin sheets, and conduct heat and electricity well. The document also discusses different groups of metals, including alkali metals, alkaline earth metals, and transition metals, and how their properties and reactivity can differ based on their position in the periodic table.