Embed presentation
Downloaded 38 times




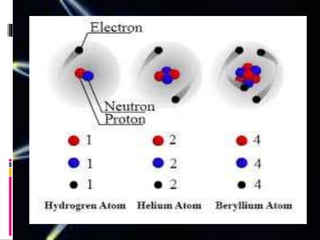

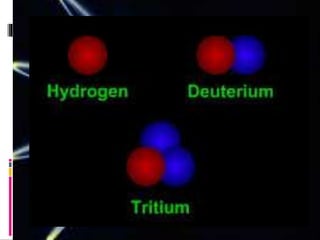





Protons, neutrons, and electrons are the three main subatomic particles. Protons have a positive charge, neutrons have no charge, and electrons have a negative charge. Isotopes are atoms of the same element that have the same number of protons but different numbers of neutrons. The atomic number identifies the number of protons in an atom's nucleus, while atomic mass refers to the total number of protons and neutrons.










