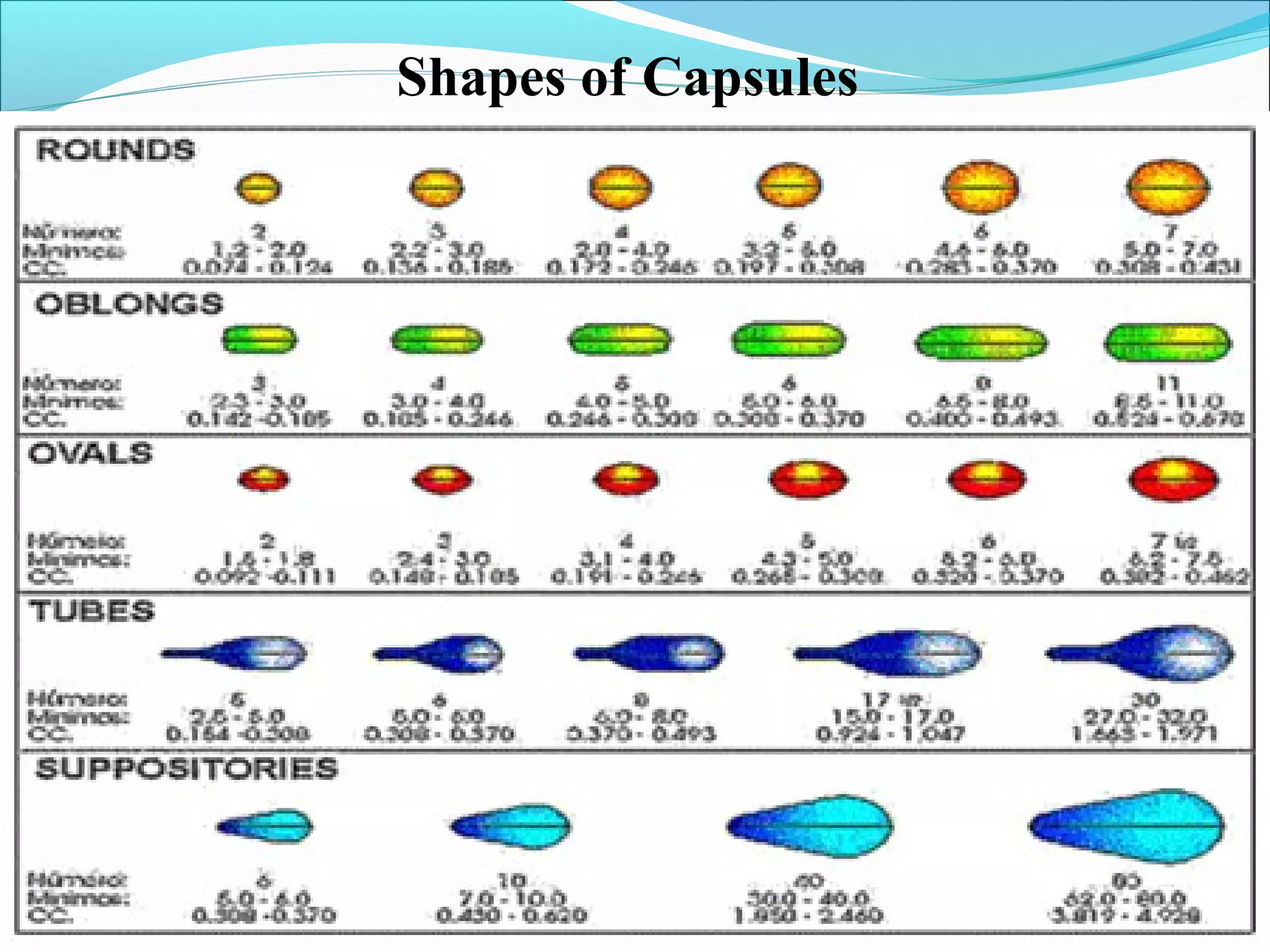
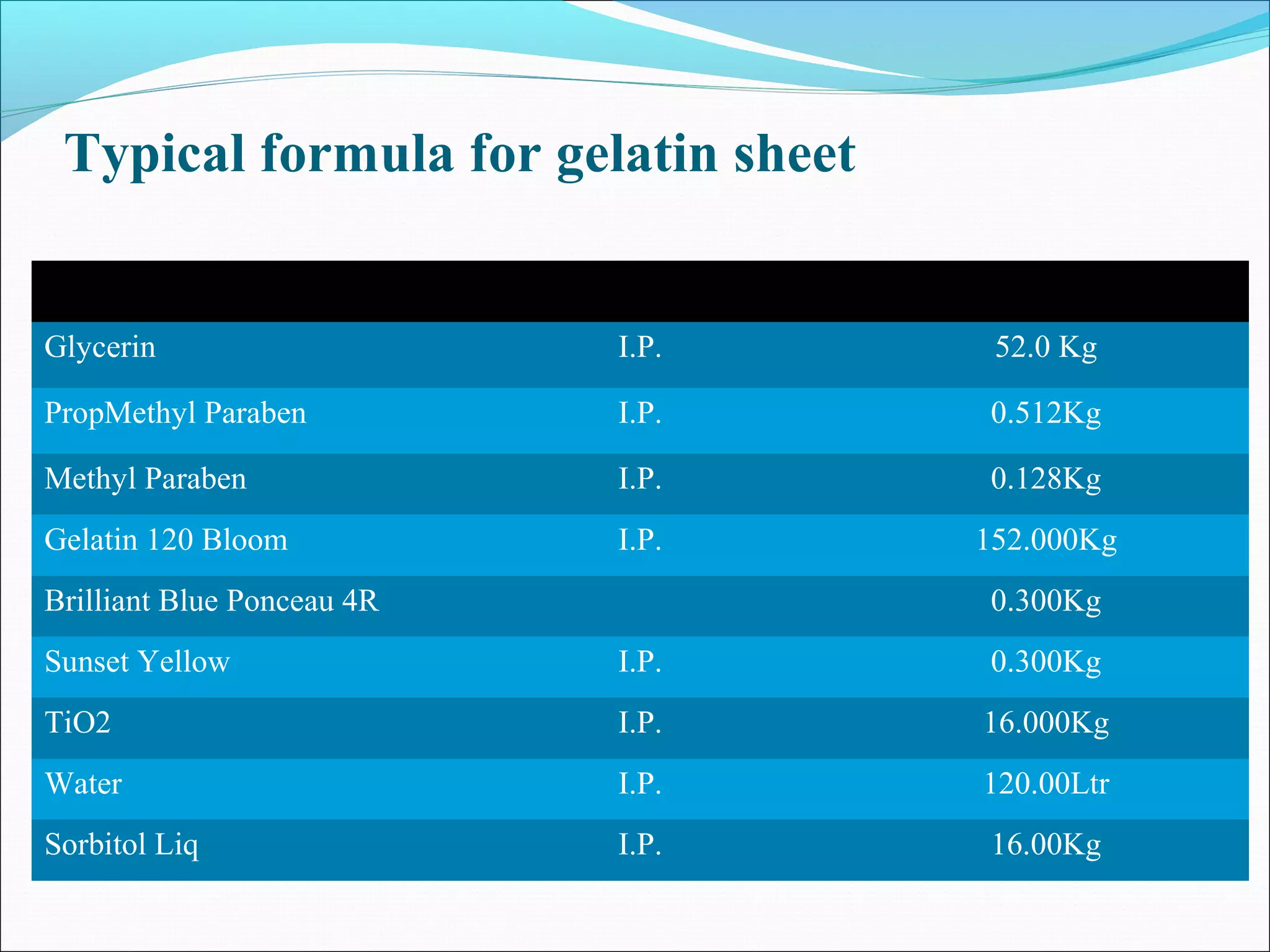
This document provides information about packing soft gelatin capsules. It discusses the advantages and disadvantages of soft gel capsules. The anatomy and composition of the capsule shell and content are described. The manufacturing process involves making the gelatin mass, filling the capsules, drying and packaging them. Quality is ensured through ingredient specifications, in-process testing of shell thickness, fill weight and moisture levels. Finished products are tested for appearance, assay, content uniformity and microbiology. Vegicaps are introduced as an animal-free alternative with benefits such as being natural and free of animal derivatives.