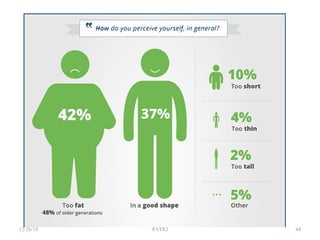
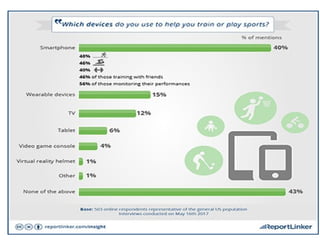
This document provides guidance for medical students on conducting health research. It outlines the key steps in the research process, including developing learning objectives and a research proposal. The proposal involves selecting a topic, reviewing existing literature, developing objectives and hypotheses, and detailing the methodology, work plan, and dissemination of results. The document also reviews best practices for writing a final research report, which consists of components like an abstract, introduction, methodology, results, discussion, and conclusions. The overall document serves to introduce medical students to the basics of the health research process.