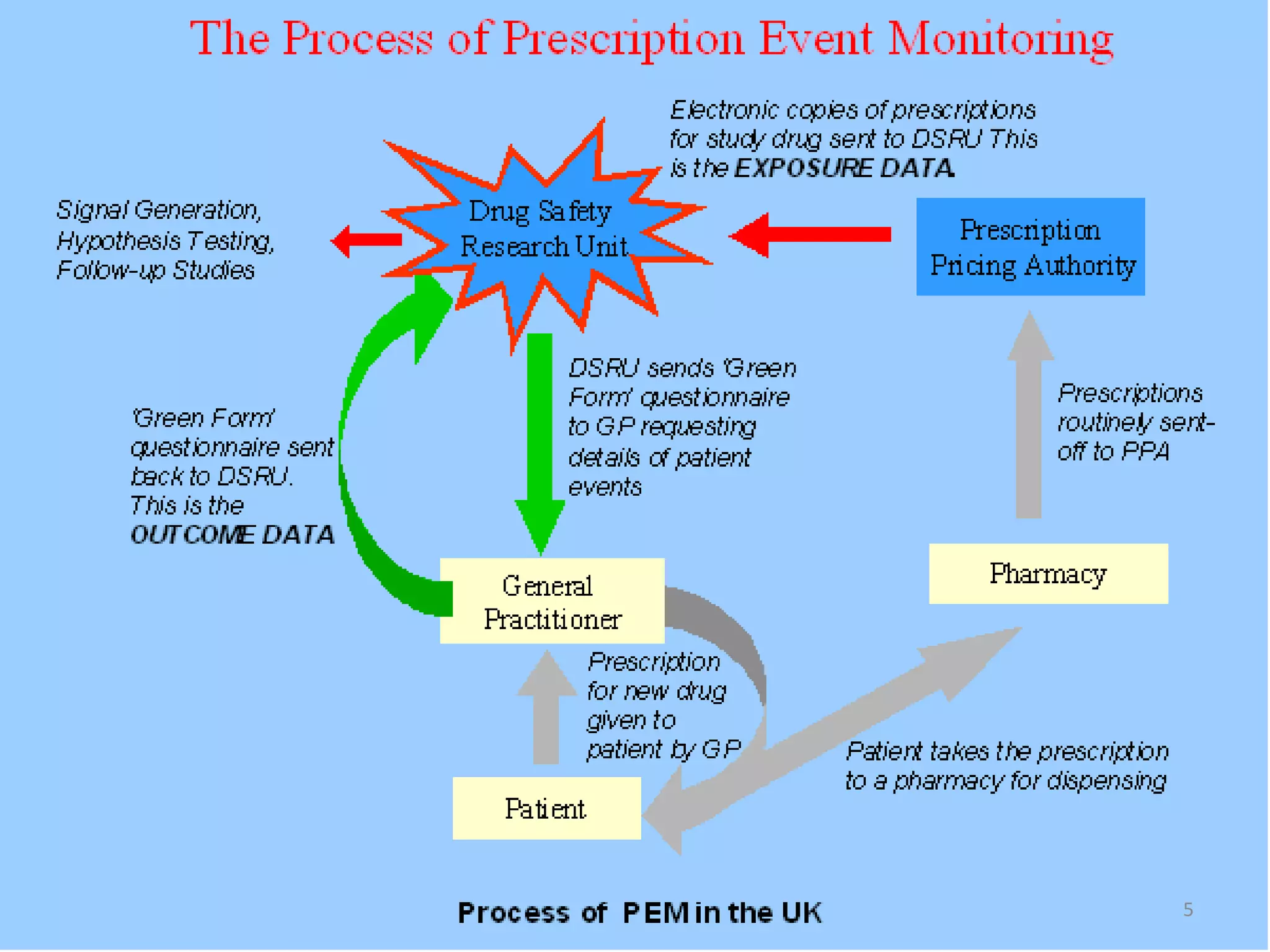
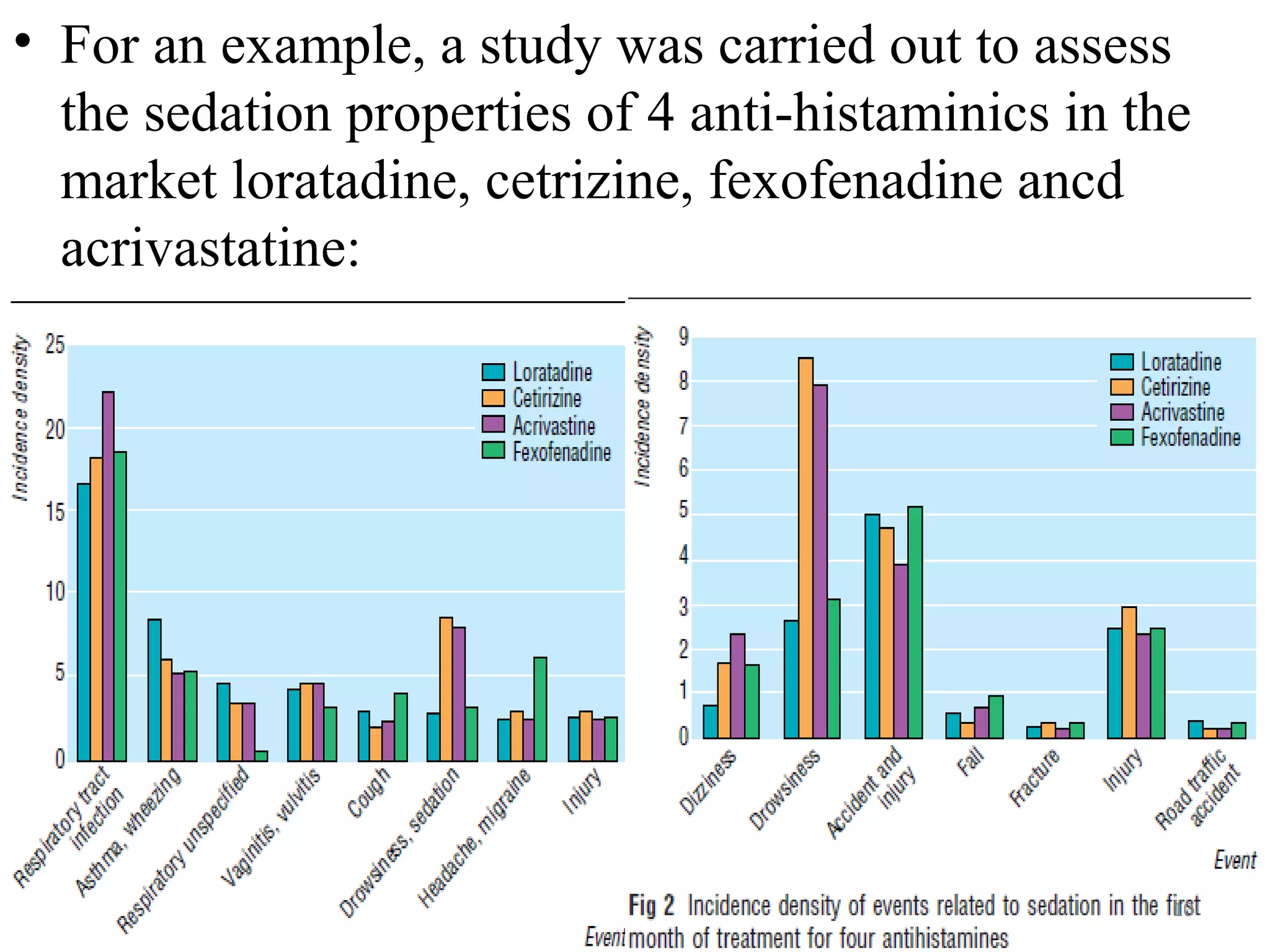
- Prescription-Event Monitoring (PEM) was established in 1981 to study the safety of new medications as used in general practice, as pre-marketing trials have limitations. PEM involves collecting data on all clinical events reported by patients after being prescribed a new drug.
- PEM provides clinically useful safety information as it establishes incidence densities of reported events from monitoring thousands of initial prescriptions of new drugs in real-world settings. This allows rare adverse drug reactions to be detected.
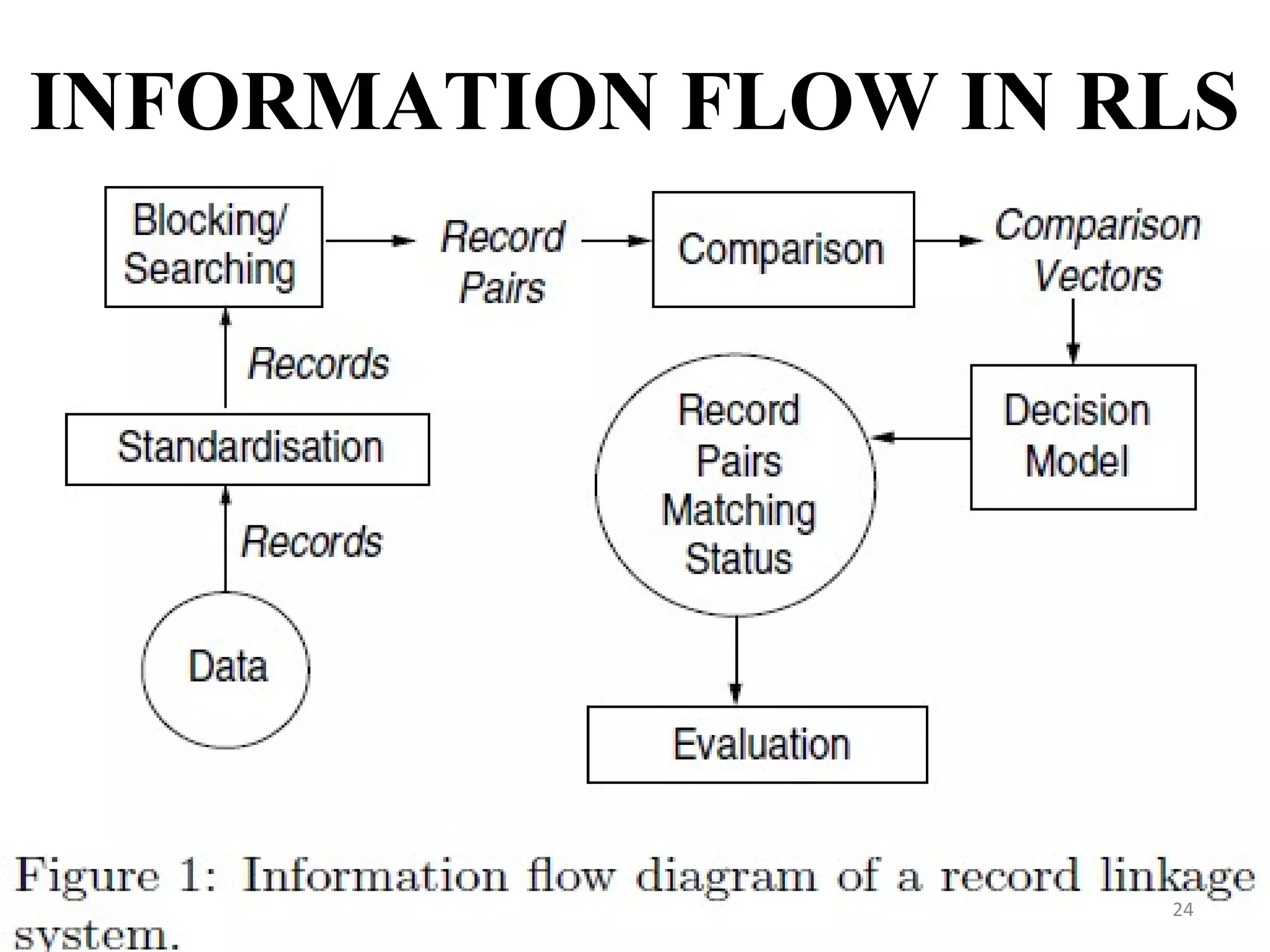
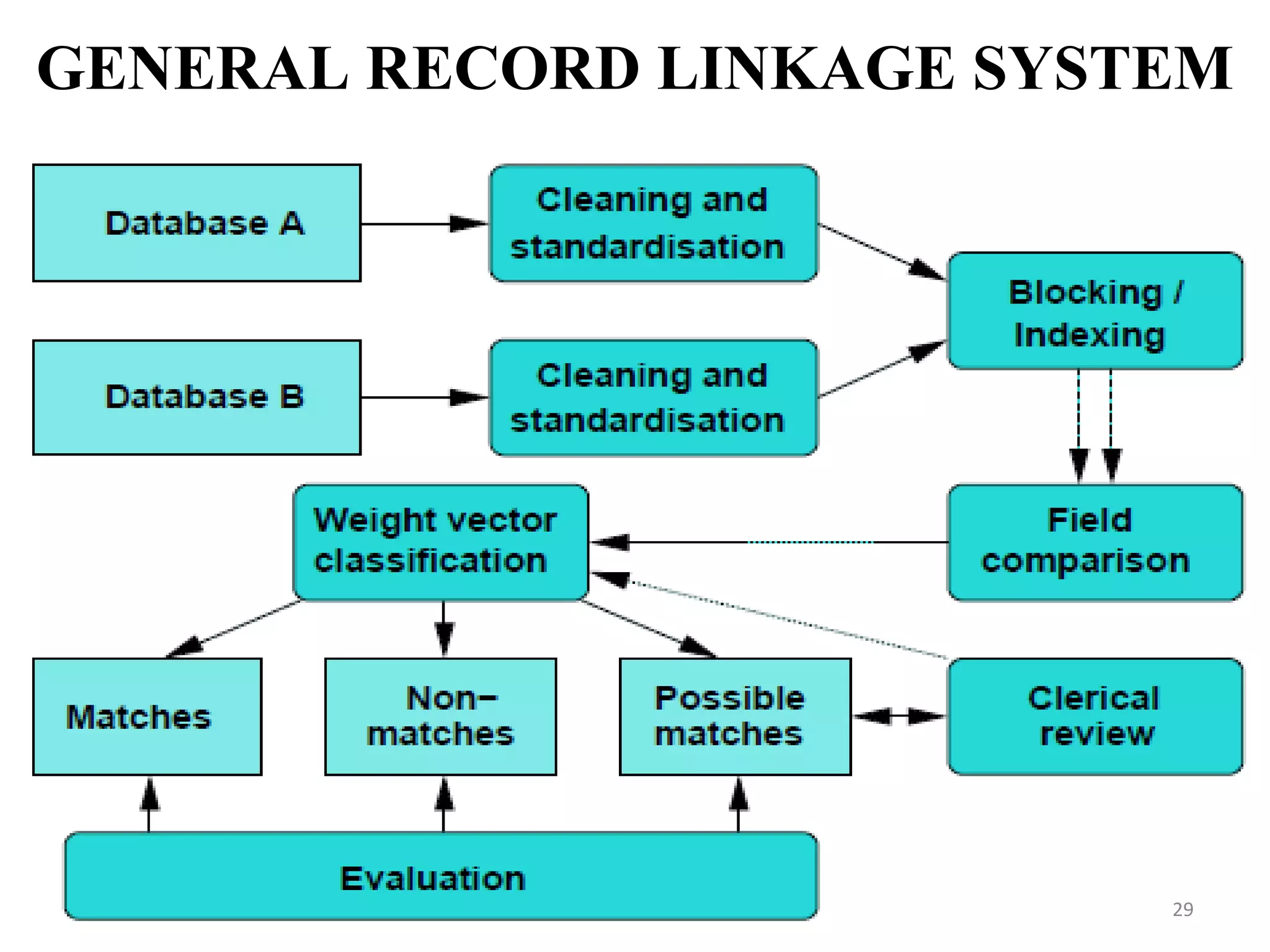
- Record linkage systems aim to link together records from different data sources that relate to the same individual or entity. This process involves standardizing, blocking, and matching records using identifiers and probabilistic methods. Record linkage improves data quality and coverage and is

















![PROBABILISTIC RECORD
LINKAGE
• Formalized by Fellegi and Sunter [1969].
• Pairs of records are classified as links, possible links, or
non-links.
• Here, we consider the probability of a match in the given
observed data.
• In probability matching, a threshold of likelihood is set
(which can be varied in different circumstances) above
which a pair of records is accepted as a match, relating to
the same person, and below which the match is rejected.
23](https://image.slidesharecdn.com/pem-rls-130422205116-phpapp02/75/Prescription-Event-Monitoring-Record-Linkage-Systems-23-2048.jpg)