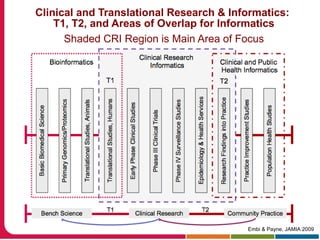
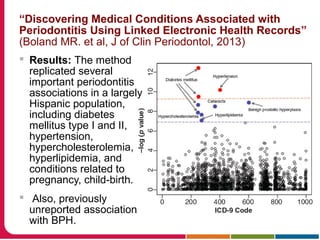
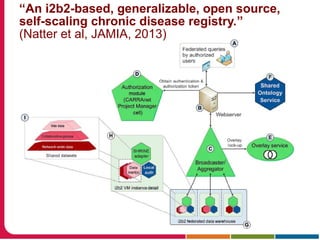
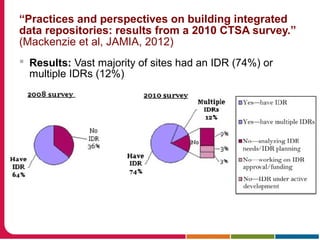
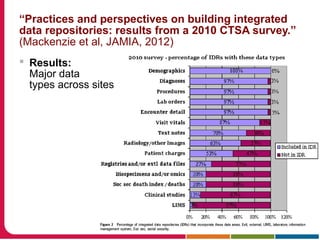
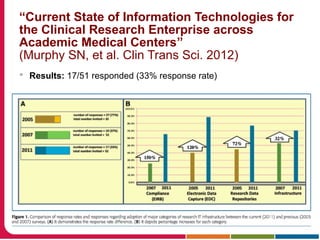
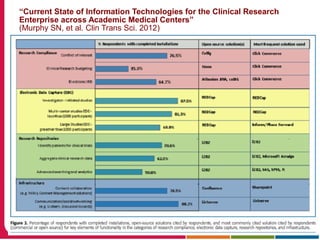
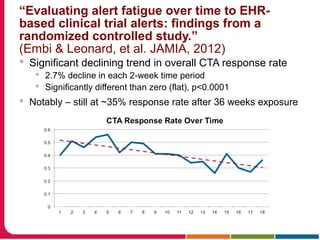
This document summarizes a presentation given by Peter Embi on clinical and translational research and informatics literature from 2012-2013. It begins with Embi's background and approach to identifying relevant papers. It then describes the topics covered in the presentation, which are grouped into categories like clinical data reuse, data management/discovery, researcher support/resources, and recruitment. For each category, 1-2 key papers are summarized in 1-3 sentences. The summaries highlight the papers' goals, methods, and conclusions. The document concludes by mentioning other notable papers and events from the past year.


![Source of Content for Session
Literature review:
Initial search by MESH terms:
("Biomedical Research"[Mesh] NOT "Genetic
Research"[Mesh]) NOT "Translational Research"[Mesh]) AND
"Informatics"[Mesh] AND "2012/01/01"[PDat] :
"2013/02/01"[Pdat]
Resulted in 90 articles; 33 were CRI relevant
Additional 483 articles found via:
Recommendations from colleagues
Other keyword searches using terms like:
Clinical Trials, Clinical Research, Informatics, Translational, Data
Warehouse, Research Registries, Recruitment
Yielding 48 more CRI-relevant articles, for a total of…
Result = 81 total CRI relevant
From those, I’ve selected 35 representative papers that
I’ll present here (briefly)](https://image.slidesharecdn.com/embicri-review-2013-final-160821054032/85/Embi-cri-review-2013-final-3-320.jpg)