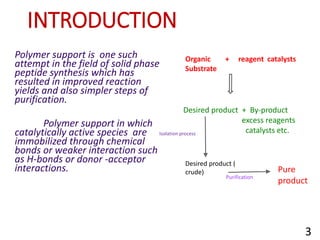
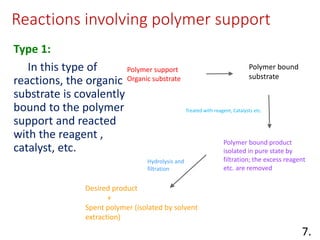
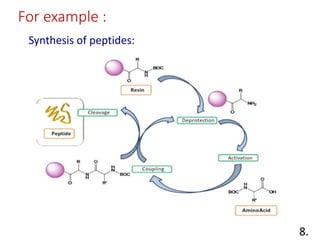
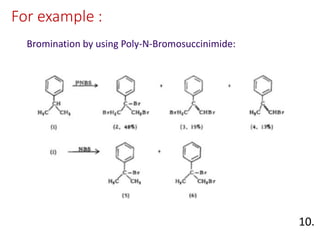
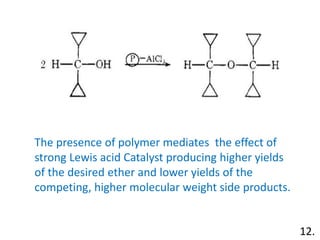
The document discusses polymer-supported synthesis in solid-phase peptide synthesis, highlighting the benefits such as improved reaction yields and simpler purification steps. It outlines properties of polymer supports, their types, and examples of reactions that utilize these supports, emphasizing the advantages of using polymers like polystyrene as catalysts. The conclusion reinforces that polymer materials enhance isolation and purification processes in organic synthesis.