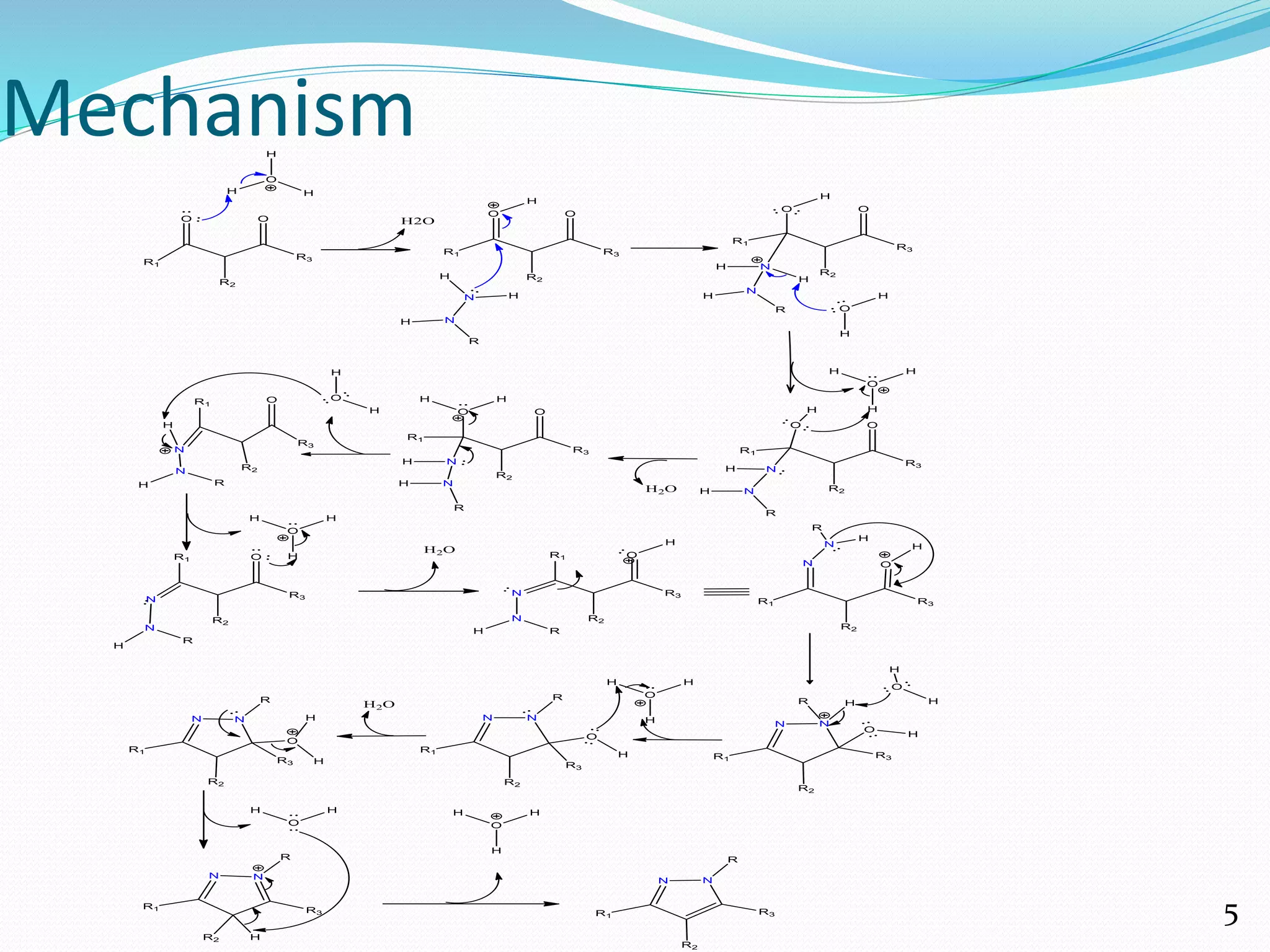
The document provides a comprehensive overview of pyrazole, a heterocyclic compound with significant medicinal properties, detailing methods of synthesis, reactions, and biological activities. Various synthesis techniques, including Knorr pyrazole synthesis and tandem reactions, are discussed along with their mechanisms. The discussion extends to pyrazole's medicinal uses, highlighting its antibacterial, anticancer, anti-inflammatory, and analgesic activities, affirming its importance as a pharmacophore with therapeutic potential.




















![Anti-Tubercular Activity
Manetti et al. identify new inhibitors of Mycobacterium
tuberculosis.
Compound (1) was found to be most active agent.
A series of 3-substituted 5-hydroxy-5-trifluoro[chloro]methyl-
1H-1-isonicotinoyl-4,5-dihydropyrazoles were synthesized by
Almeida da Silva et al. and tested for their in vitro antimicrobial
activity against Mycobacterium tuberculosis H37Rv, INH-
resistant clinical M.tuberculosis isolates non tuberculous
mycobacteria.
22](https://image.slidesharecdn.com/annu-200606065230/75/Heterocyclic-compounds-pyrazole-22-2048.jpg)



