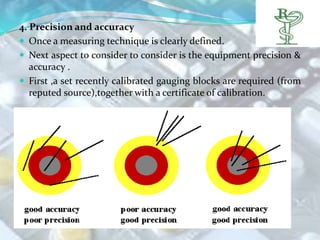
This document discusses key aspects of pharmaceutical packaging design and specifications. It begins by introducing packaging as integral to new product development and quality control. It then covers types of packaging, objectives of packaging, and why standardized designs are important for regulatory compliance and production efficiency. The document outlines important considerations for packaging design like marketing involvement, validation trials, and customer usability. It also discusses component specifications, quality testing standards, and layout of specification documents. Overall, the document provides a comprehensive overview of pharmaceutical packaging design and specifications from conceptualization through regulatory requirements.