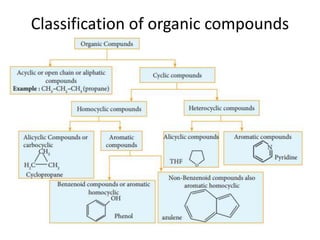
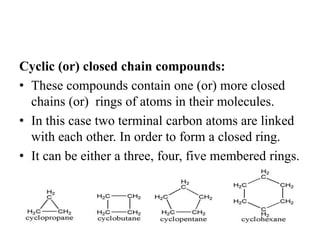
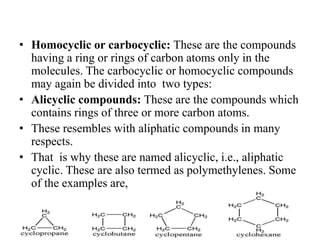
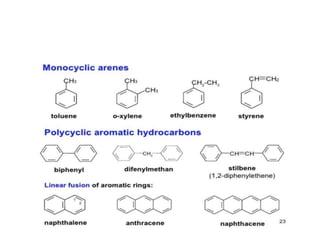
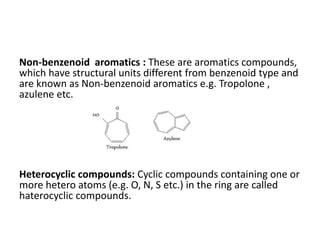
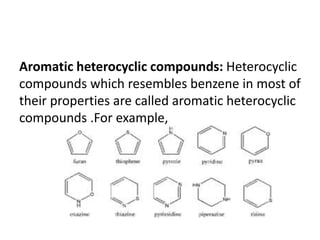
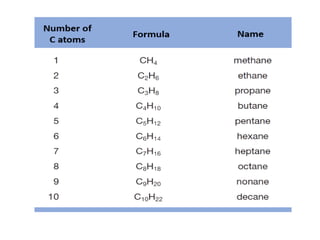
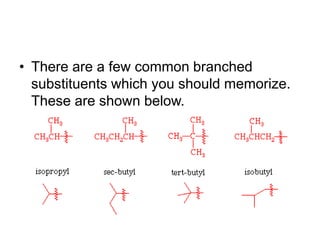
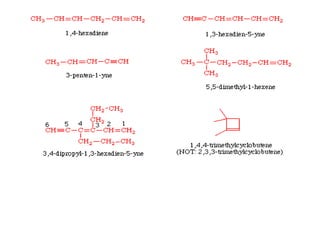
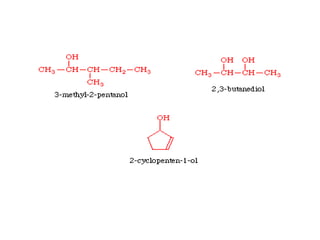
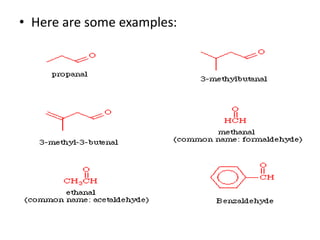
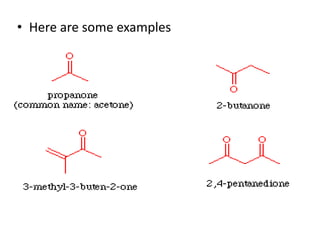
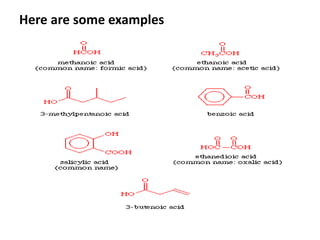
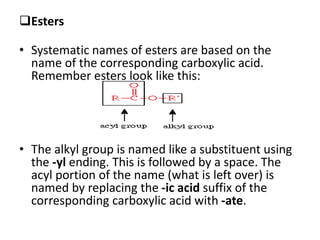
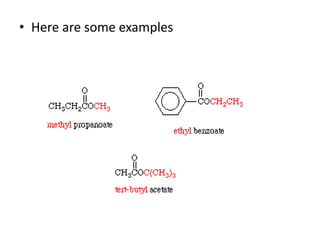
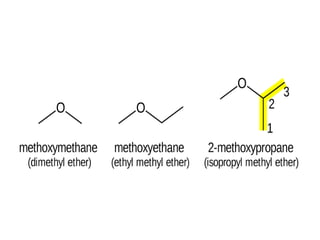
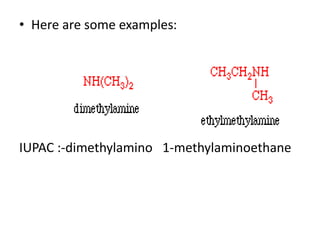
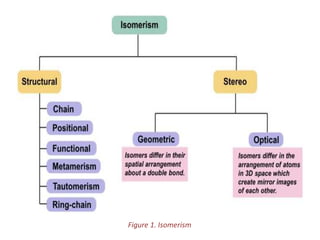
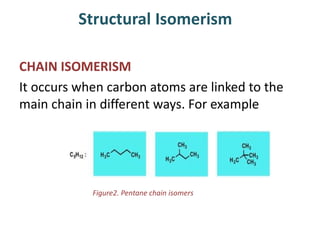
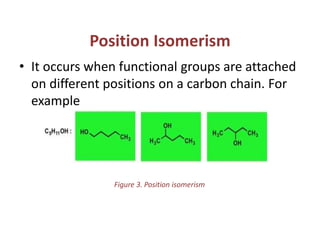
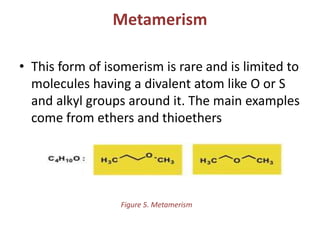
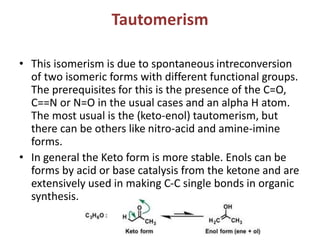
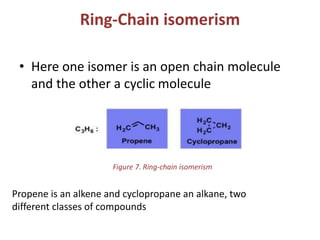
This document provides information on the classification, nomenclature, and isomerism of organic compounds. It discusses the classification of organic compounds as acyclic, cyclic, alicyclic, aromatic, heterocyclic, and more. It also covers IUPAC nomenclature rules for naming alkanes, alkenes, alkynes, alkyl halides, alcohols, aldehydes, ketones, carboxylic acids, esters, and ethers. Examples are provided to illustrate the nomenclature rules for different functional groups. Structural isomerism in organic compounds is also briefly mentioned.