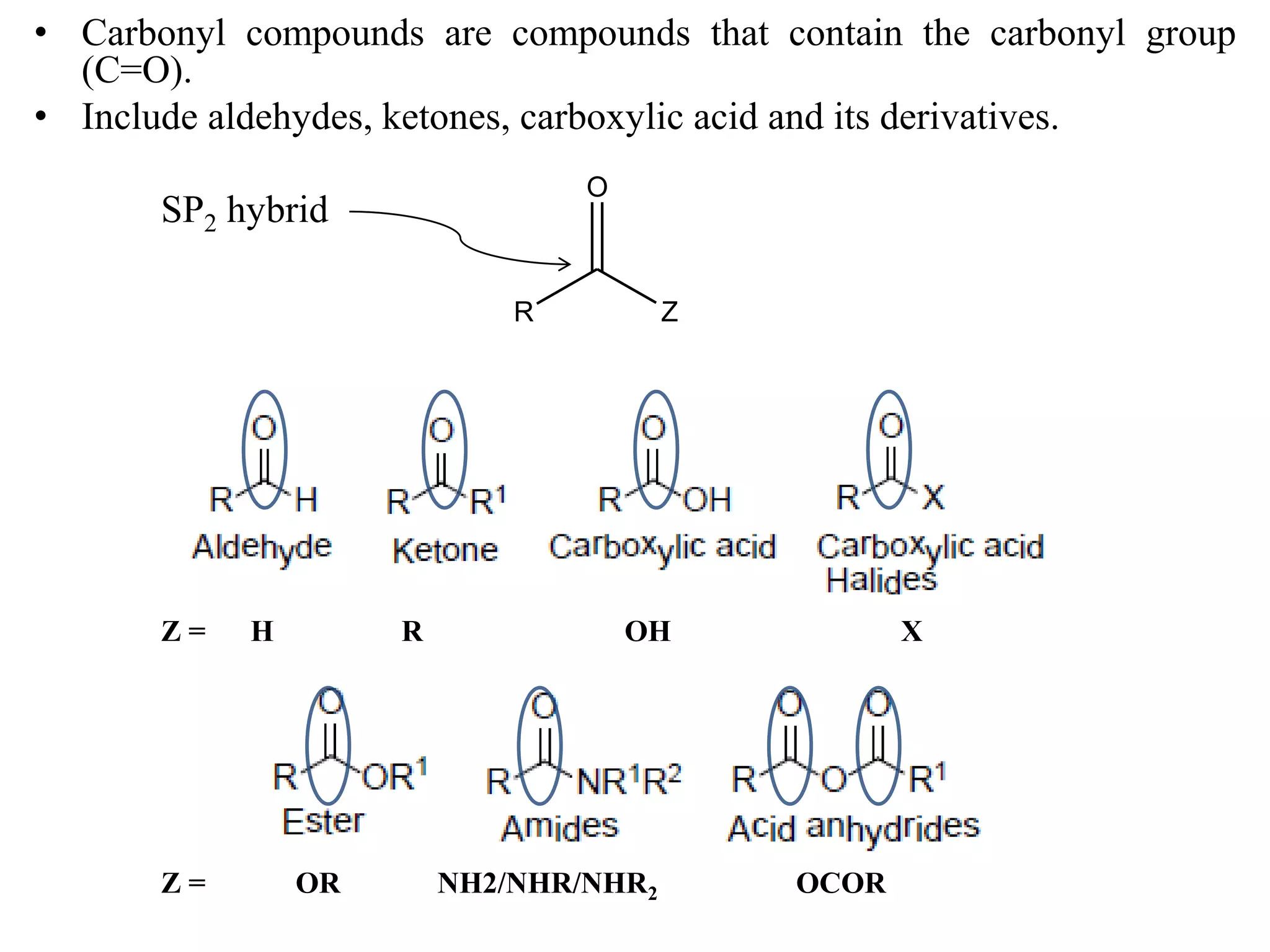
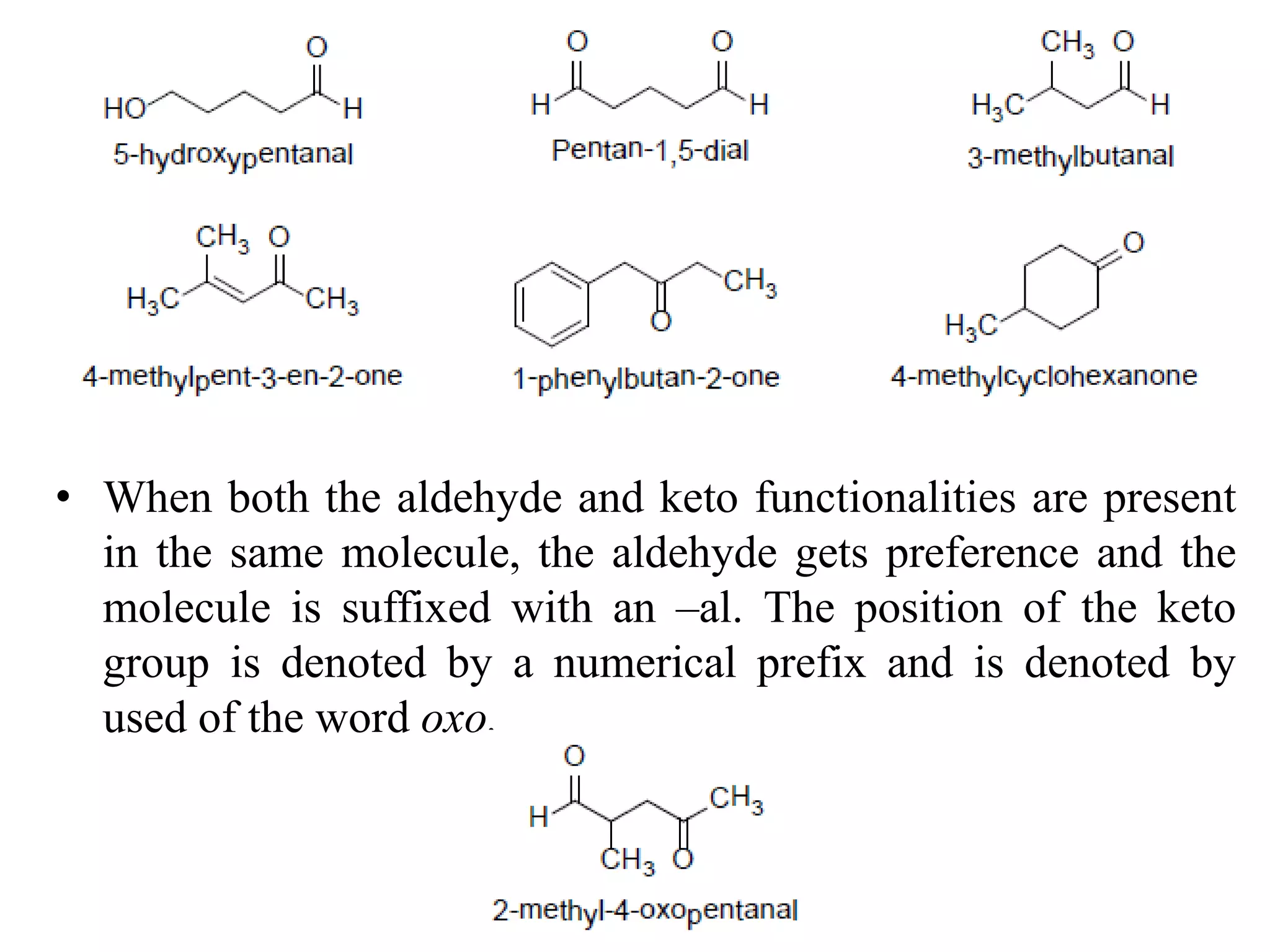
This document provides information about carbonyl compounds, specifically aldehydes and ketones. It discusses their IUPAC nomenclature, methods of preparation including oxidation of alcohols and oxidative cleavage of alkenes, and physical and chemical properties. The chemical reactions covered include nucleophilic addition, reduction, condensation, and oxidation reactions. Examples of important aldehydes and ketones are also mentioned along with their structures and uses.