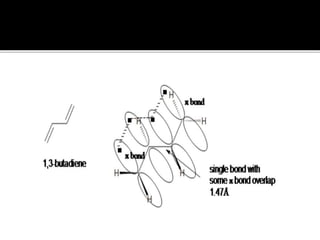
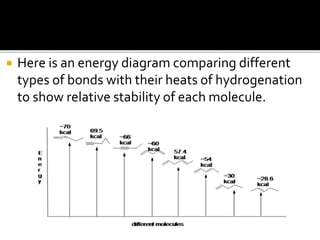
Conjugated dienes contain alternating double and single carbon bonds. The single bond between conjugated double bonds is shorter than a typical alkane bond due to delocalization of pi electrons across the carbon chain. This delocalization through resonance structures makes conjugated dienes more stable than non-conjugated dienes. The extra stability of conjugated dienes is also due to the lower hybridization energy from sp2 hybridized carbons compared to the sp3 carbons in non-conjugated dienes.