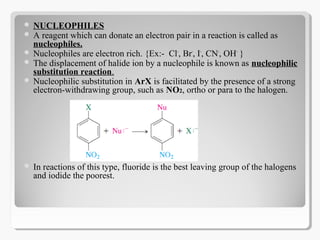
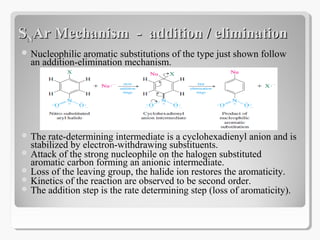
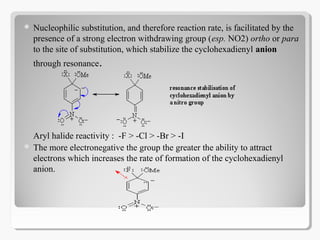
Nucleophilic aromatic substitution reactions follow an addition-elimination mechanism known as SNAr. The rate-determining step is the formation of a cyclohexadienyl anion intermediate through nucleophilic attack. Electron-withdrawing groups stabilize this intermediate through resonance, making the reaction faster. Nucleophilic aromatic substitution is most favorable when the leaving group is fluoride and least with iodide, and occurs readily with strong nucleophiles like hydroxide or cyanide in the presence of electron-withdrawing groups ortho or para to the reaction site.