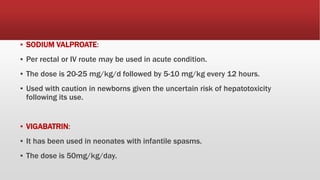
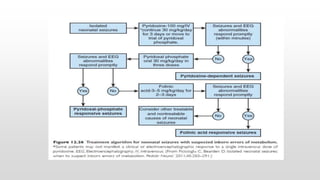
This document provides an overview of neonatal seizures. Key points include:
- Neonatal seizures have an incidence of 10.3 per 1000 live births and are more common in preterm infants.
- They are defined as abnormal excessive neuronal activity causing alterations in motor, behavioral or autonomic functions.
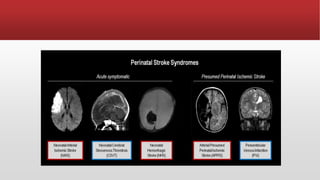
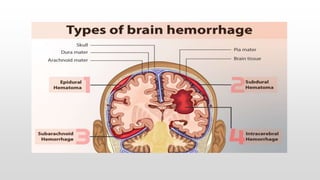
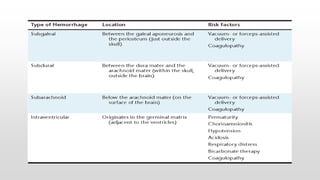
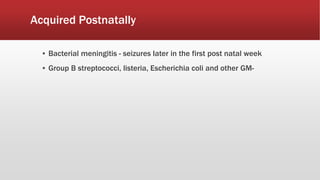
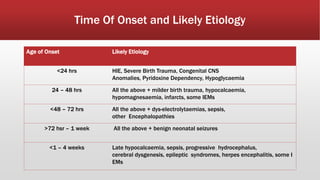
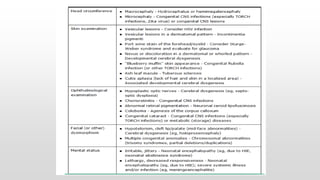
- Common causes include hypoxic-ischemic encephalopathy, structural brain lesions, infections, and metabolic disturbances.
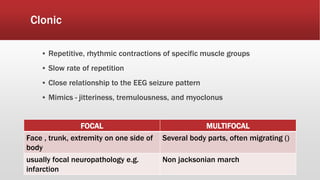
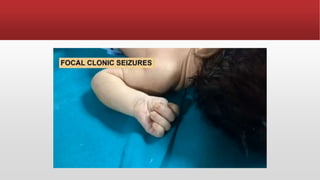
- Seizures are classified based on their clinical presentation and EEG findings as subtle, clonic, tonic, myoclonic or EEG-only.
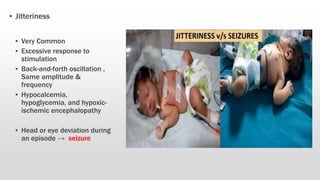
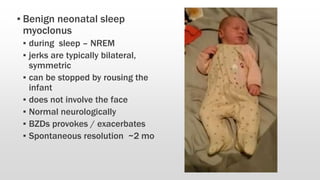
- Differentiating seizures from non-seizure events like apnea or jitteriness is important.












































































![REFERENCES
1. Cloherty and starks manual of neonatal care 8th edition.
2. Aiims protocols in neonatology
3. Avery’s diseases of the newborn. -- 9th ed. / [Edited by] christine
A. Gleason, sherin U. Devask
4. Fanaroff and martin’s neonatal-perinatal medicine : diseases of
the fetus and infant / [edited by] richard J. Martin, avroy A.
Fanaroff, michele C. Walsh.—10th edition.](https://image.slidesharecdn.com/neonatalseizuredrpraman3-210713035852/85/Neonatal-seizure-by-dr-praman-98-320.jpg)