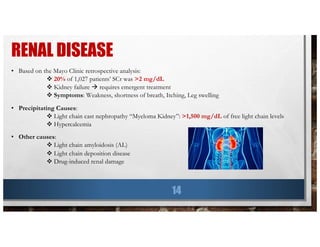
This document provides information about multiple myeloma, including key statistics, risk factors, symptoms, diagnosis, and tests. It summarizes findings from a retrospective analysis of over 1,000 multiple myeloma patients at the Mayo Clinic from 1985 to 1988. The analysis found that the median age at diagnosis was 66 years old, common symptoms at presentation included anemia (73% of patients), bone pain (60% of patients), and renal disease (20% of patients had elevated creatinine). The median survival time was 33 months and did not improve from 1985 to 1988.












![NEUROLOGICAL DISEASES
16
• Radiculopathy:
v Most common neurological complication of Multiple Myeloma
v Thoracic/lumbosacral area
v Compression of nerve by paravertebral plasmacytoma or collapsed bone
• Cord Compression:
v 5% of patients
v Compression by extramedullary plasmacytoma or bone fragment of vertebral bone fracture
v Symptoms: Severe back pain with weakness, lower extremity paresthesias, bladder/bowel dysfunction
• Peripheral Neuropathy “Pins and Needles”:
v Uncommon in Multiple Myeloma
v If present, due to amyloidosis
• CNS Involvement:
v Intracranial plasmacytomas are rare
v Poor prognosis: Leptomeningeal myelomatosis (Lactate dehydrogenase [LDH] levels ↑)
v Rare: Encephalopathy due to hyperviscosity or high blood levels of ammonia](https://image.slidesharecdn.com/multiplemyeloma-190726081006/85/Multiple-Myeloma-16-320.jpg)



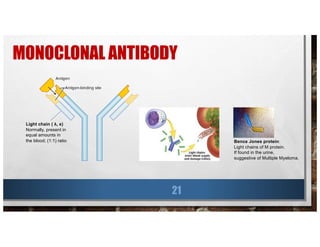



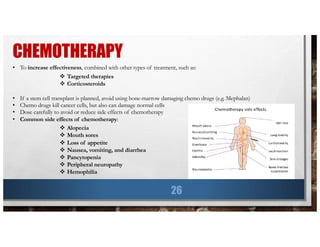





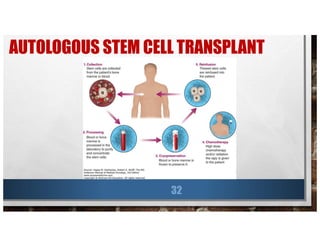
![LOCAL THERAPY
33
• Treats cancer tumor without affecting the rest of the body
• More useful for earlier stage (less advanced) cancers
• Types:
v Surgery: Used to relief symptoms of MM, does NOT treat the disease
Spinal cord compressions à paralysis, severe muscle weakness, numbness
Remove single plasmacytomas
Prevent or treat bone fractures with metal plates and rods
v Radiation: Use of high-energy rays or particles to kill cancer cells
Eases bone pain unresponsive to other drugs
Prevents paralysis in spinal cord collapse
External beam radiation therapy: machine outside the body
Side effects: skin changes in treated area (redness, peeling, blistering), fatigue,
nausea, diarrhea, low blood counts [SEs go away once treatment is over]](https://image.slidesharecdn.com/multiplemyeloma-190726081006/85/Multiple-Myeloma-33-320.jpg)


