This document provides information about stroke, including:
1. Stroke is caused by a lack of oxygen to the brain from blocked or ruptured arteries, and is a leading cause of death and disability in the US and worldwide.
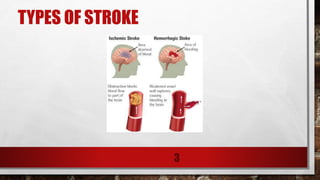
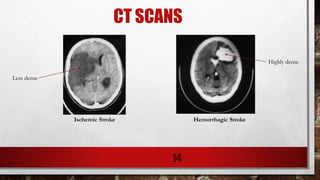
2. The two main types of stroke are ischemic (87% of cases) and hemorrhagic (13% of cases).
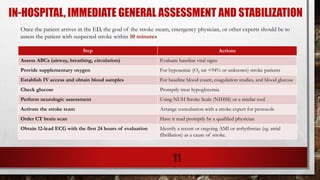
3. Signs of stroke include sudden weakness, confusion, trouble speaking, vision issues, loss of balance, and severe headaches with no known cause. Early treatment leads to better outcomes.

























![PATIENT CASE
Laboratory @1:26 pm 3-15-18:
Sodium, Serum: 140 [135 - 145 mmol/L]
Potassium, Serum: 3.9 [3.5 - 5.3 mmol/L]
Chloride, Serum: 109 [96 - 108 mmol/L]
Carbon Dioxide, Serum: 27 [22 - 31 mmol/L]
Anion Gap, Serum: 4 [5 - 17 mmol/L]
Blood Urea Nitrogen, Serum: 12 [7 - 23 mg/dL]
Creatinine, Serum: 0.73 [0.50 - 1.30 mg/dL]
Glucose, Serum: 87 [70 - 99 mg/dL]
Calcium, Total Serum: 9.3 [8.5 - 10.1 mg/dL]
Protein Total, Serum: 7.8 [6.0 - 8.3 gm/dL]
Albumin, Serum: 4.1 [3.3 - 5.0 g/dL]
Bilirubin Total, Serum: 0.4 [0.2 - 1.2 mg/dL]
Alkaline Phosphatase, Serum: 104 [40 - 120 U/L]
Aspartate Aminotransferase (AST/SGOT): 21 [15 - 37 U/L]
Alanine Aminotransferase (ALT/SGPT): 29 [12 - 78 U/L]
eGFR if Non African American: 96 [>=60 mL/min/1.73M2]
eGFR if African American: 111 [>=60 mL/min/1.73M2]
Creatinine Kinase, Serum: 77 [26 - 192 U/L]
Troponin I, Serum: <0.015 [0.015 - 0.045 ng/mL]
Activated Partial Thromboplastin Time: 30.6 [27.5 - 37.4 sec]
Prothrombin Time, Plasma: 11.0 [9.8 - 12.7 sec]
31](https://image.slidesharecdn.com/acutestroke-190726080550/85/Acute-Stroke-31-320.jpg)
![PATIENT CASE
Hematology @1:26 pm 3-15-18:
WBC Count: 4.86 [3.80 - 10.50 K/uL]
RBC Count: 5.14 [3.80 - 5.20 M/uL]
Hemoglobin: 16.0 [11.5 - 15.5 g/dL]
Hematocrit: 48.4 [34.5 - 45.0 %]
Mean Cell Volume: 94.2 [80.0 - 100.0 fl]
Mean Cell Hemoglobin: 31.1 [27.0 - 34.0 pg]
Mean Cell Hemoglobin Conc: 33.1 [32.0 - 36.0 gm/dL]
Red Cell Distrib Width: 14.3 [10.3 - 14.5 %]
Platelet Count - Automated: 233 [150 - 400 K/uL]
Auto Neutrophil #: 2.52 [1.80 - 7.40 K/uL]
Auto Lymphocyte #: 1.55 [1.00 - 3.30 K/uL]
Auto Monocyte #: 0.62 [0.00 - 0.90 K/uL]
Auto Eosinophil #: 0.10 [0.00 - 0.50 K/uL]
Auto Basophil #: 0.05 [0.00 - 0.20 K/uL]
Auto Neutrophil %: 51.8 [43.0 - 77.0 %]
Auto Lymphocyte %: 31.9 [13.0 - 44.0 %]
Auto Monocyte %: 12.8 [2.0 - 14.0 %]
Auto Eosinophil %: 2.1 [0.0 - 6.0 %]
Auto Basophil %: 1.0 [0.0 - 2.0 %]
Auto Immature Granulocyte %: 0.4 [0.0 - 1.5 %]
Nucleated RBC: 0 [0 - 0 /100 WBCs]
32](https://image.slidesharecdn.com/acutestroke-190726080550/85/Acute-Stroke-32-320.jpg)









