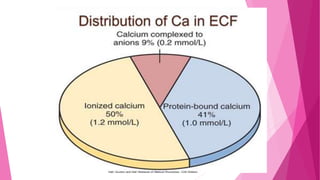
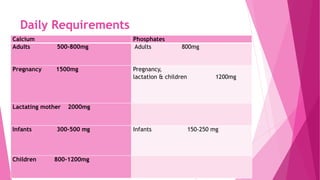
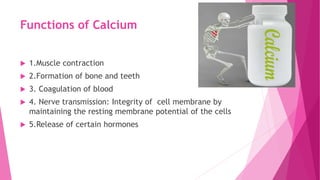
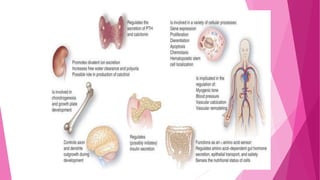
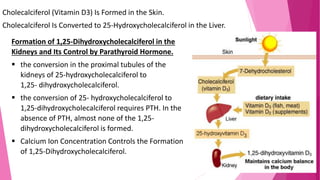
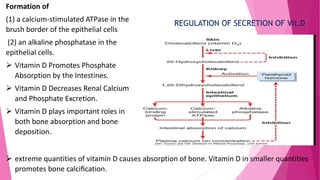
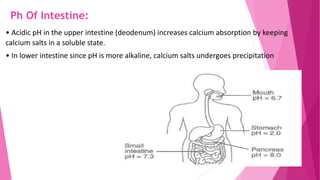
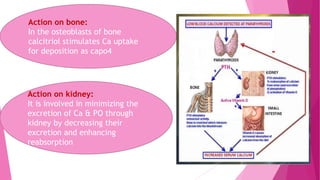
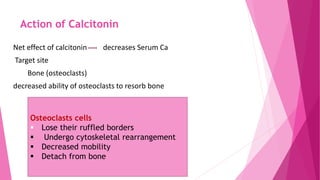
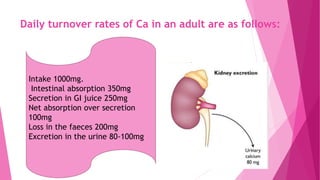
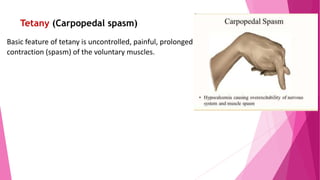
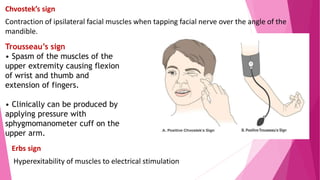
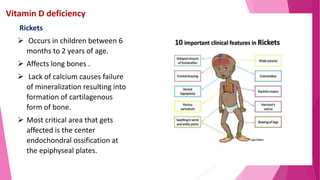
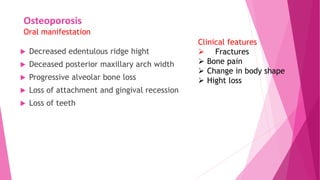
The document summarizes key information about calcium and phosphorus metabolism. It discusses their daily requirements, distribution in the body, dietary sources, functions, factors controlling absorption such as vitamin D, parathyroid hormone, and calcitonin. It also outlines hormonal control of calcium and phosphorus metabolism and clinical importance of hypo- and hypercalcemia and hyperphosphatemia. The objectives are to understand the role of calcium and phosphorus in the body and factors influencing their metabolism.