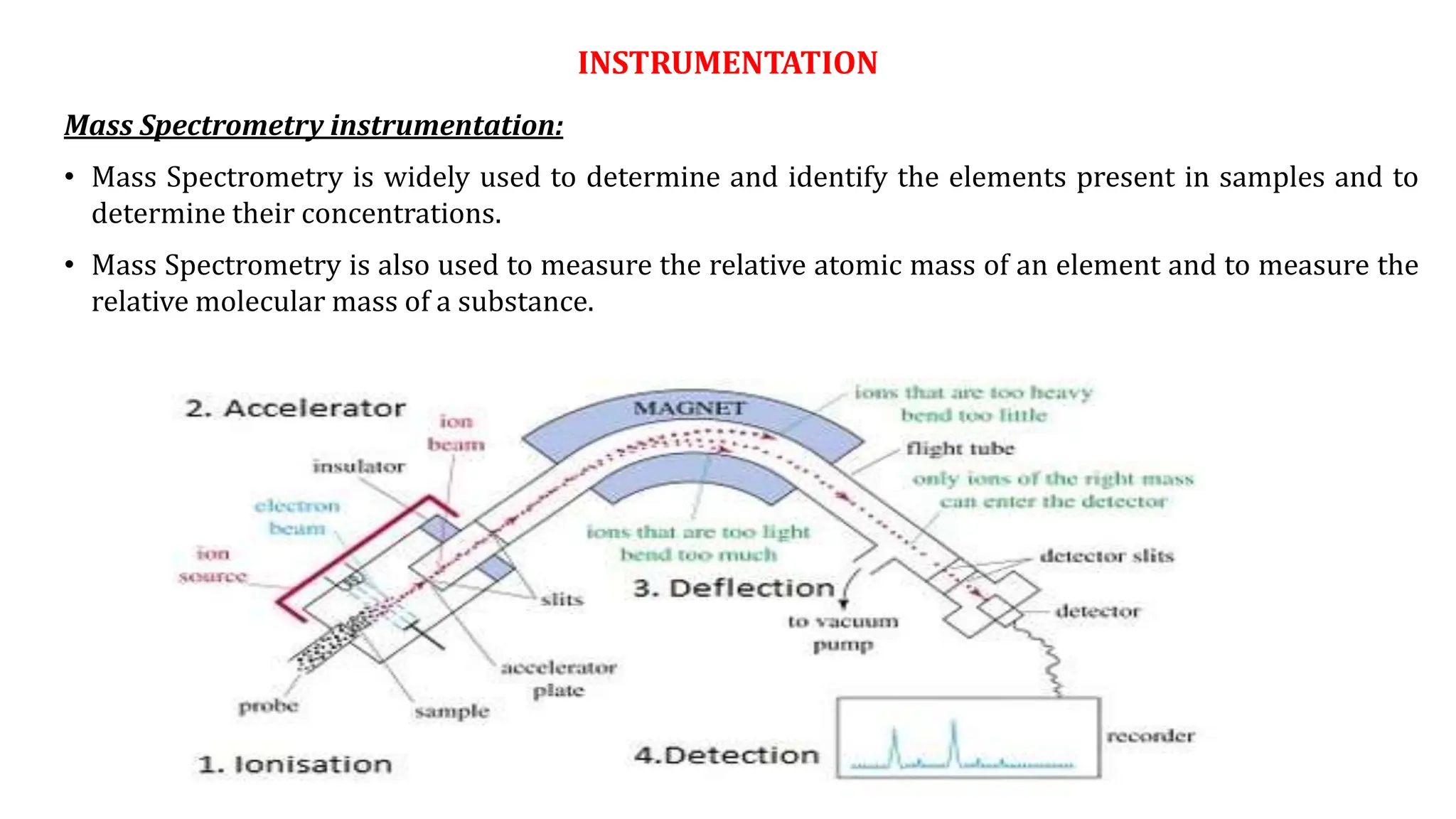
Mass spectrometry is a technique that uses electron bombardment to convert neutral molecules into positively charged ions without using electromagnetic radiation. The positively charged ions are then separated based on their mass-to-charge ratios using electric or magnetic fields. The sample is bombarded with electrons, which knocks electrons off the molecules to make them positively charged ions. The ions are then accelerated and deflected by electric and magnetic fields based on their mass, with lighter ions deflected more. When the ions hit a detector, their signals are amplified and converted into a mass spectrum that identifies the elements or molecules in the sample. Mass spectrometry is widely used for elemental analysis and molecular identification across various scientific fields.