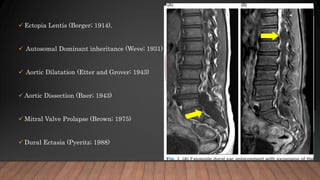
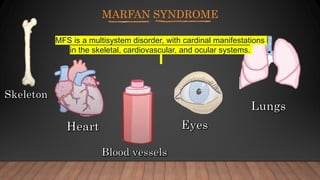
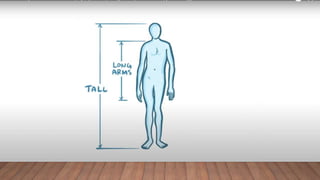
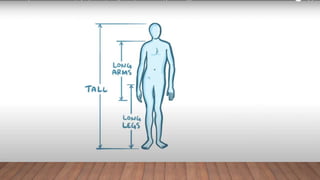
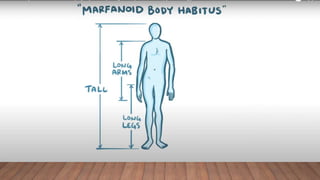
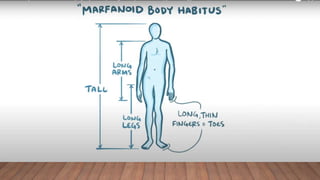
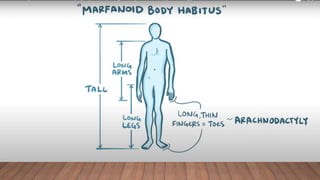
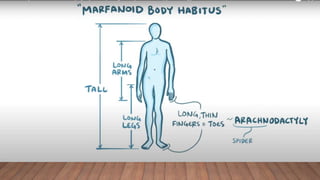
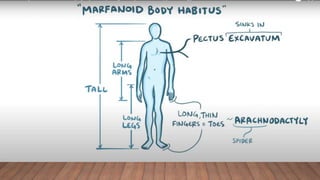
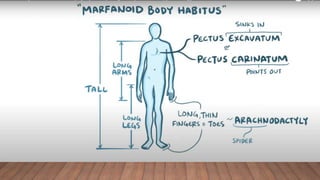
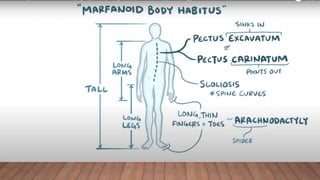
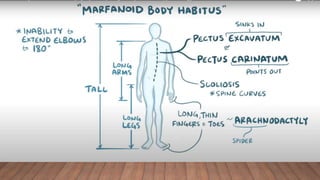
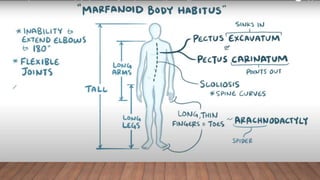
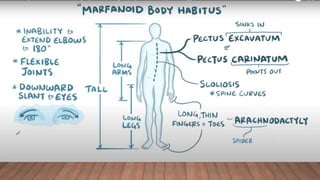
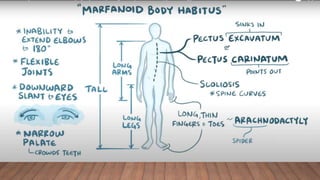
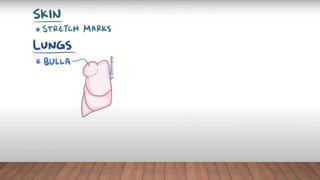
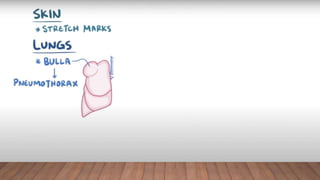
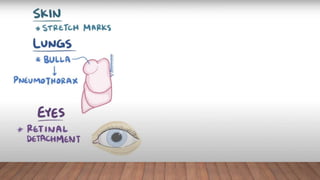
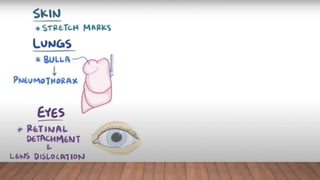
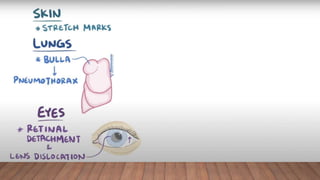
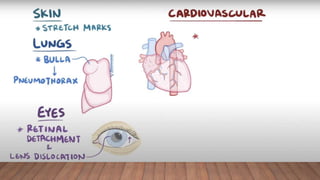
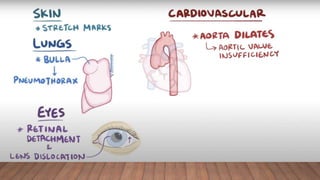
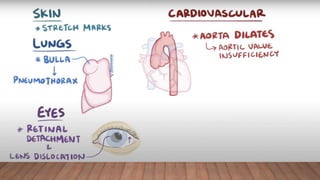
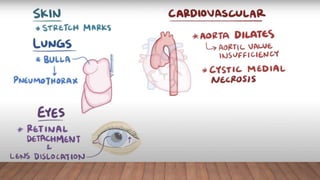
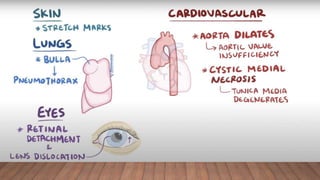
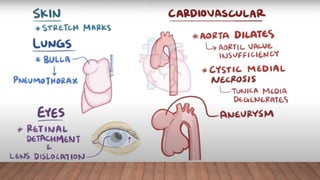
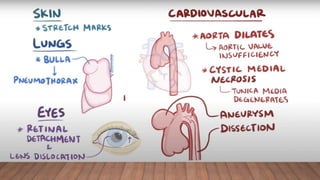
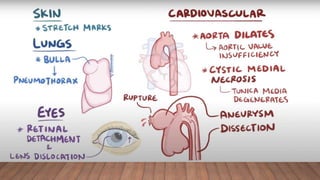
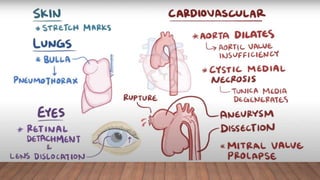
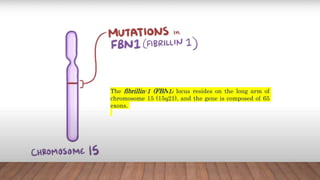
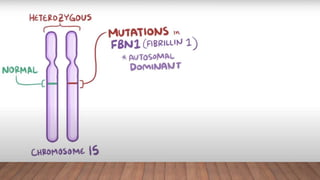
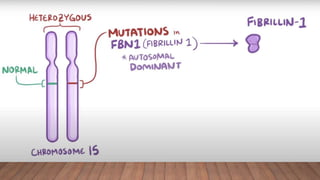
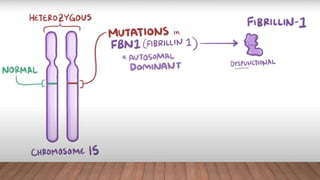
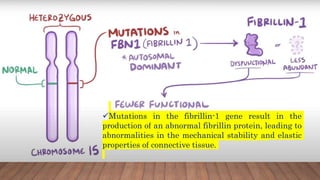
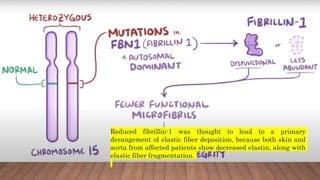
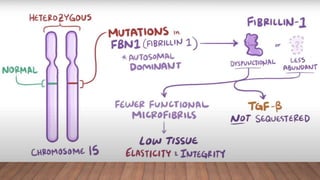
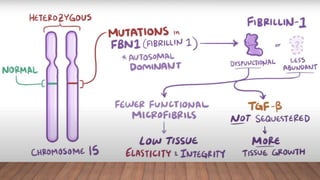
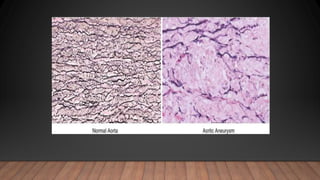
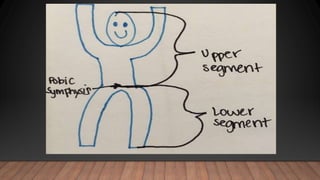
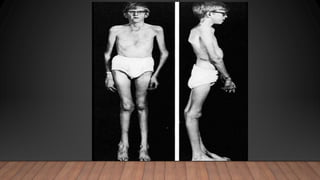
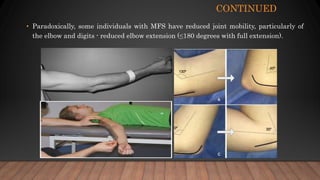
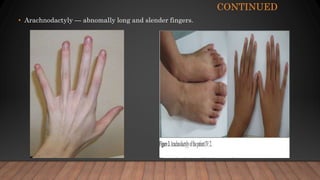
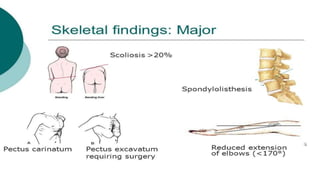
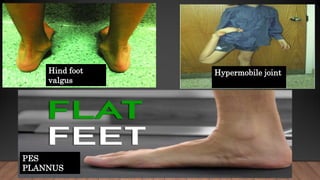
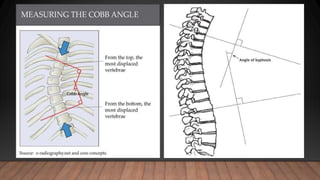
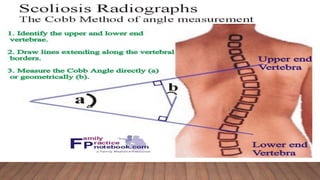
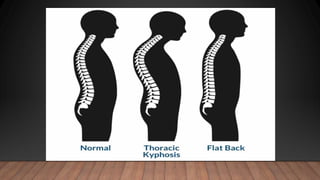
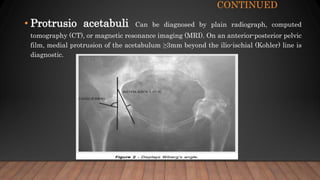
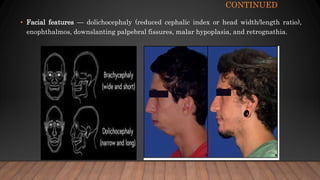
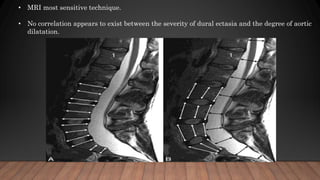
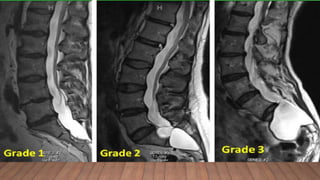
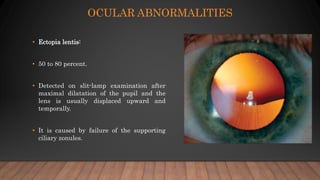
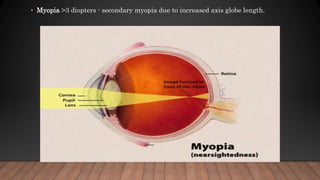
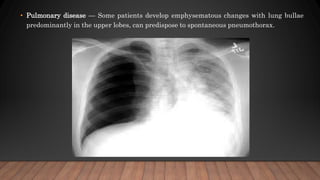
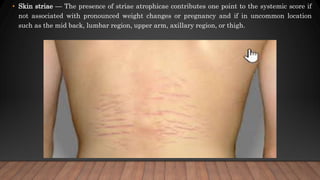
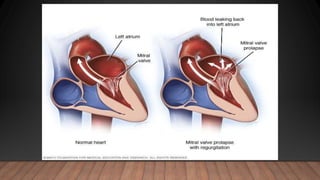
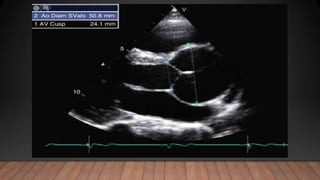
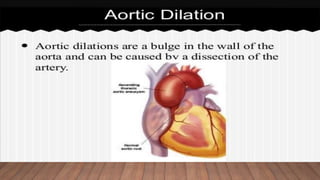
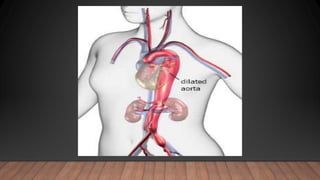
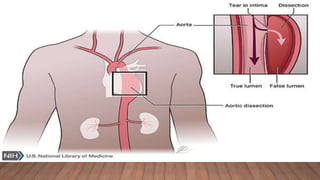
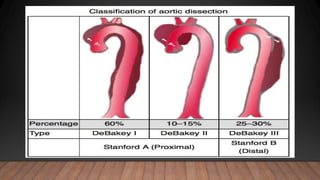
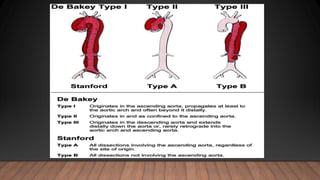
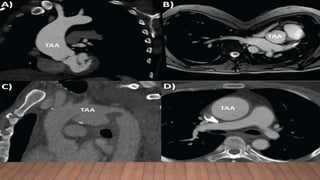
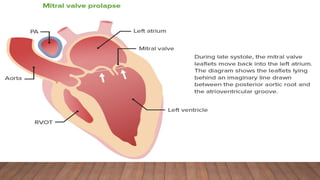
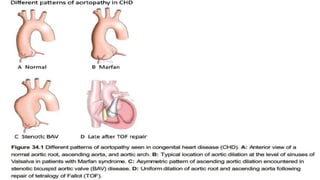
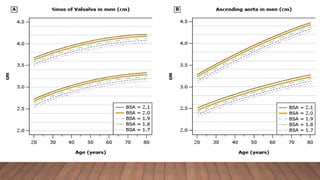
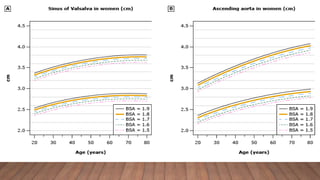
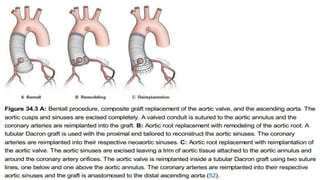
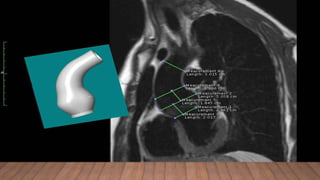
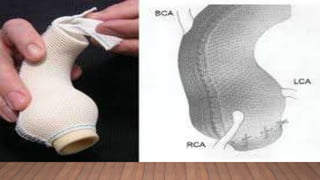
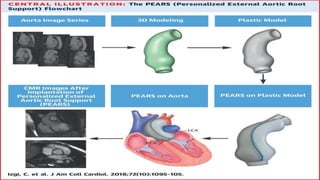
Marfan syndrome is a genetic disorder of connective tissue caused by mutations in the FBN1 gene. It affects the skeletal, ocular, and cardiovascular systems. Key features include disproportionately long limbs, joint laxity, eye lens dislocation, and aortic root aneurysm/dissection which are the leading causes of mortality. Diagnosis is based on clinical criteria involving multiple organ systems. Ongoing monitoring of the aorta is critical as progressive dilatation can lead to dissection.