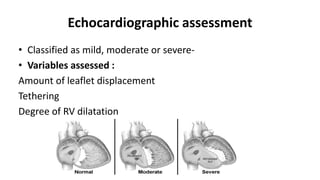
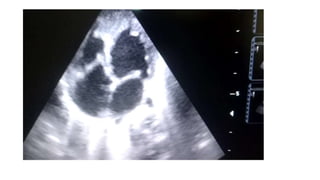
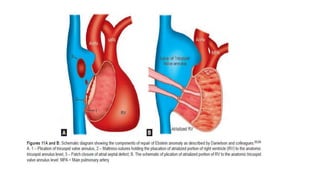
This document provides an overview of Ebstein's anomaly, a congenital heart defect where the tricuspid valve is displaced downward into the right ventricle. It discusses the history, anatomy, pathophysiology, clinical presentation, diagnosis using echocardiography, associated abnormalities, and surgical management. Surgical techniques include biventricular repair to reconstruct the tricuspid valve, single ventricle palliation with right ventricle exclusion, and cardiac transplantation for the most severe cases with significant biventricular dysfunction.