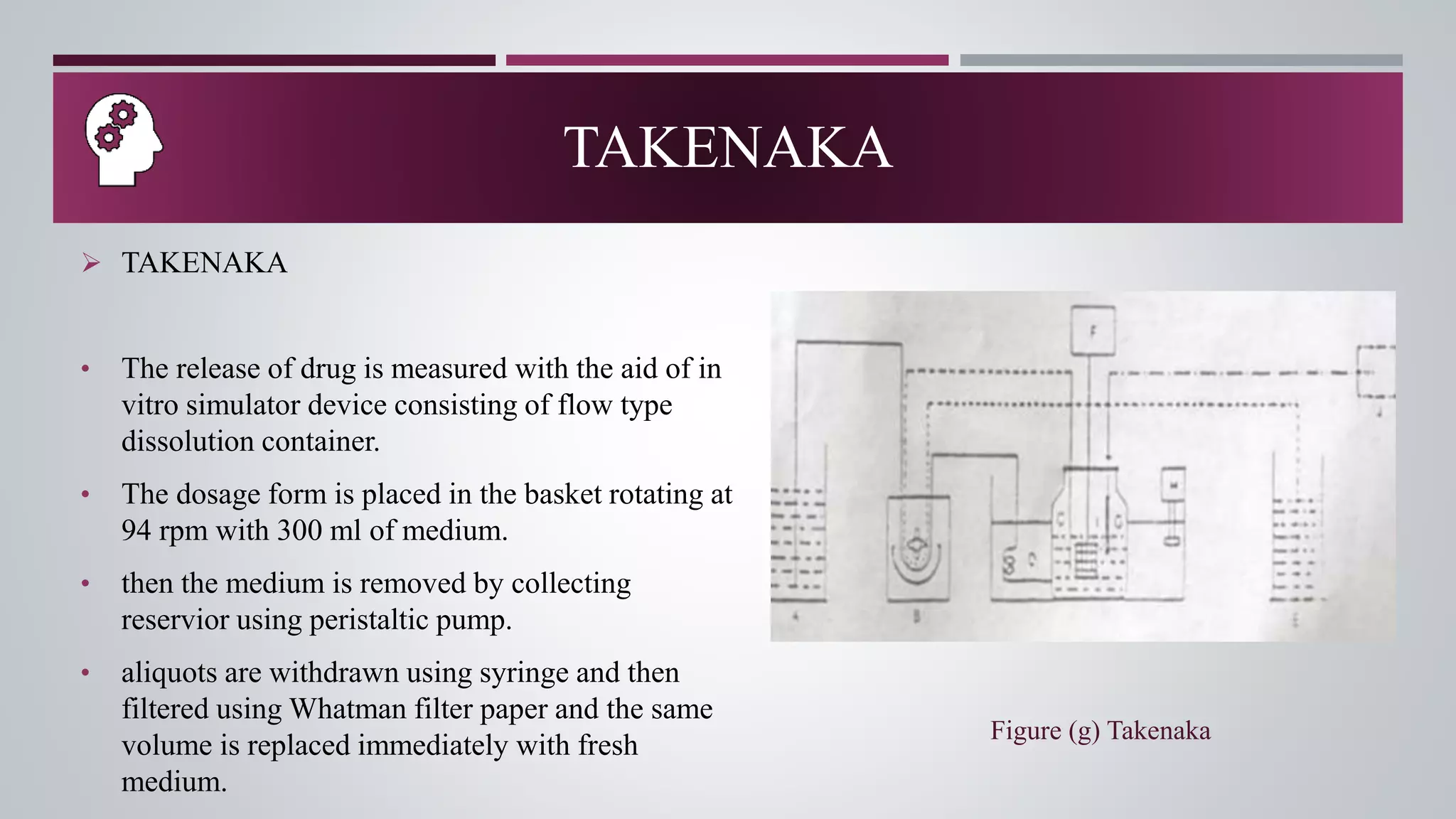
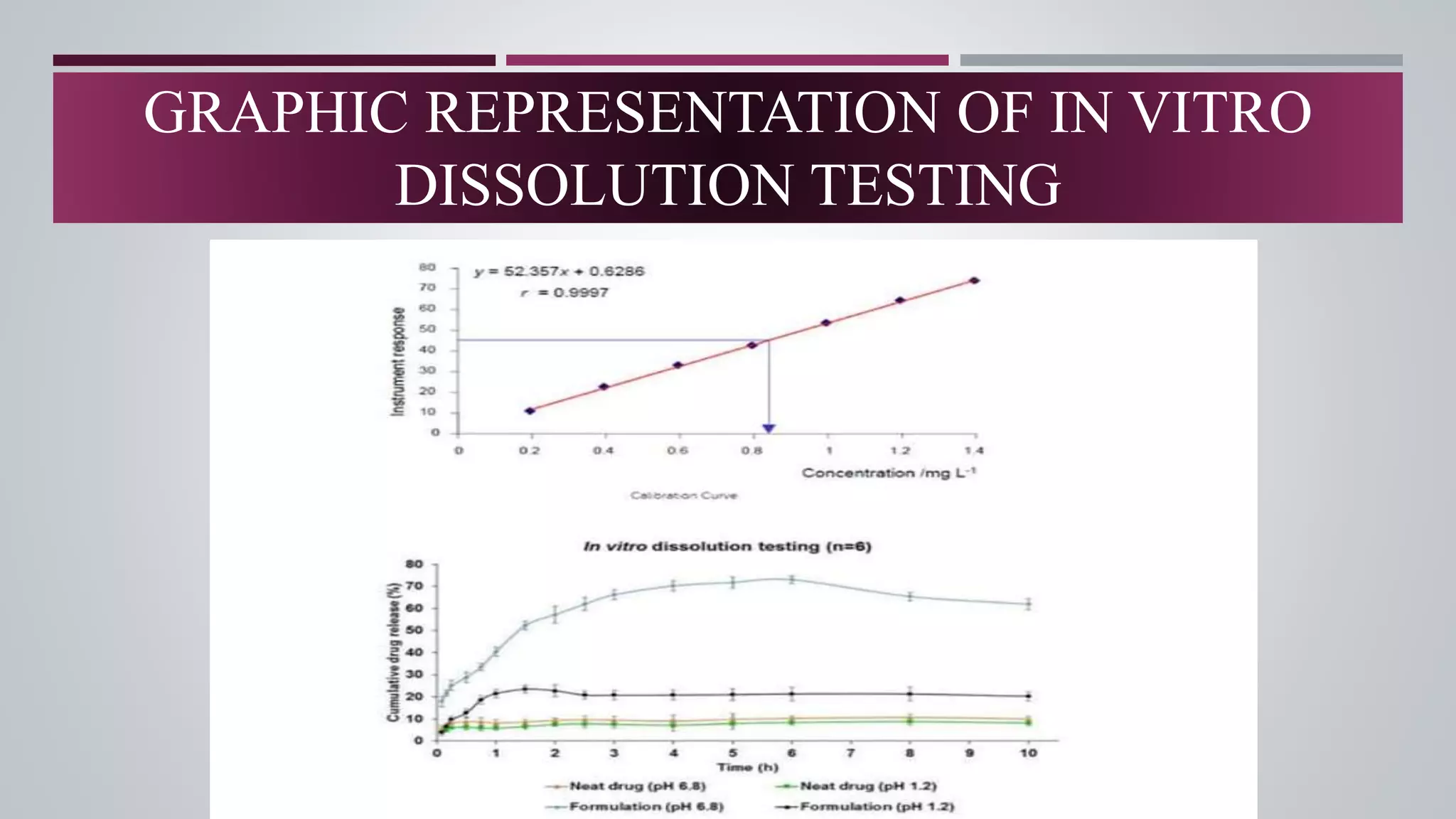
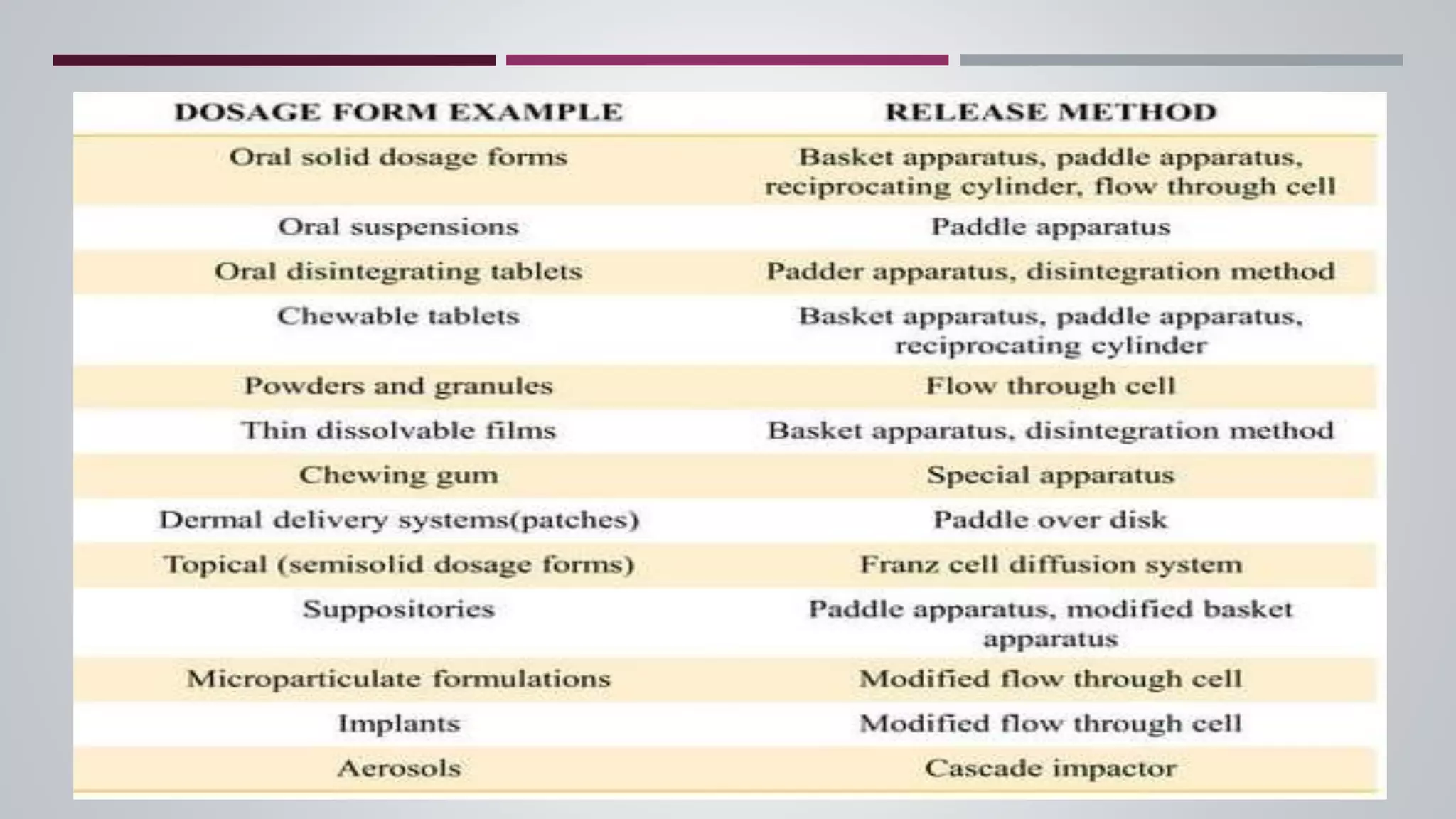
The document discusses in vitro dissolution and drug release testing, highlighting its importance for assessing the performance of drug products regarding bioavailability and therapeutic effectiveness. It outlines various dissolution methods, including non-sink and sink techniques, and describes specific methodologies like the Klein solvmeter, Nelson hanging pellet method, and rotating flask apparatus. Additionally, it covers the classifications of drug release such as immediate, sustained, and controlled release, along with in vitro testing procedures used to evaluate drug release profiles.