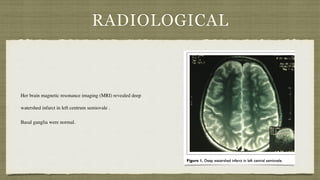
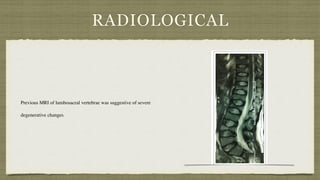
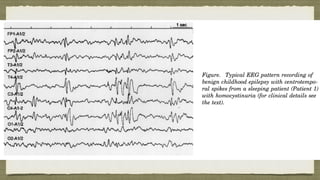
This document describes two case reports of patients with atypical benign childhood epilepsy with centrotemporal spikes (BECTS) who were also diagnosed with homocystinuria.
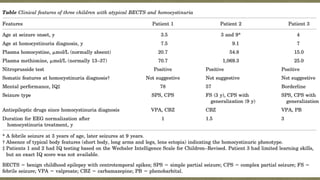
Patient 1 experienced seizures characterized by right arm jerking and speech arrest from age 3.5. Testing at age 6 showed BECTS on EEG but homocystinuria was not diagnosed until 18 months later. Treatment for homocystinuria resolved her seizures.
Patient 2 had a single febrile seizure at age 3 and two afebrile seizures at age 9 with EEG also showing BECTS. He was later found to have homocystinuria biochemically. Both patients showed improvement of neurological symptoms with