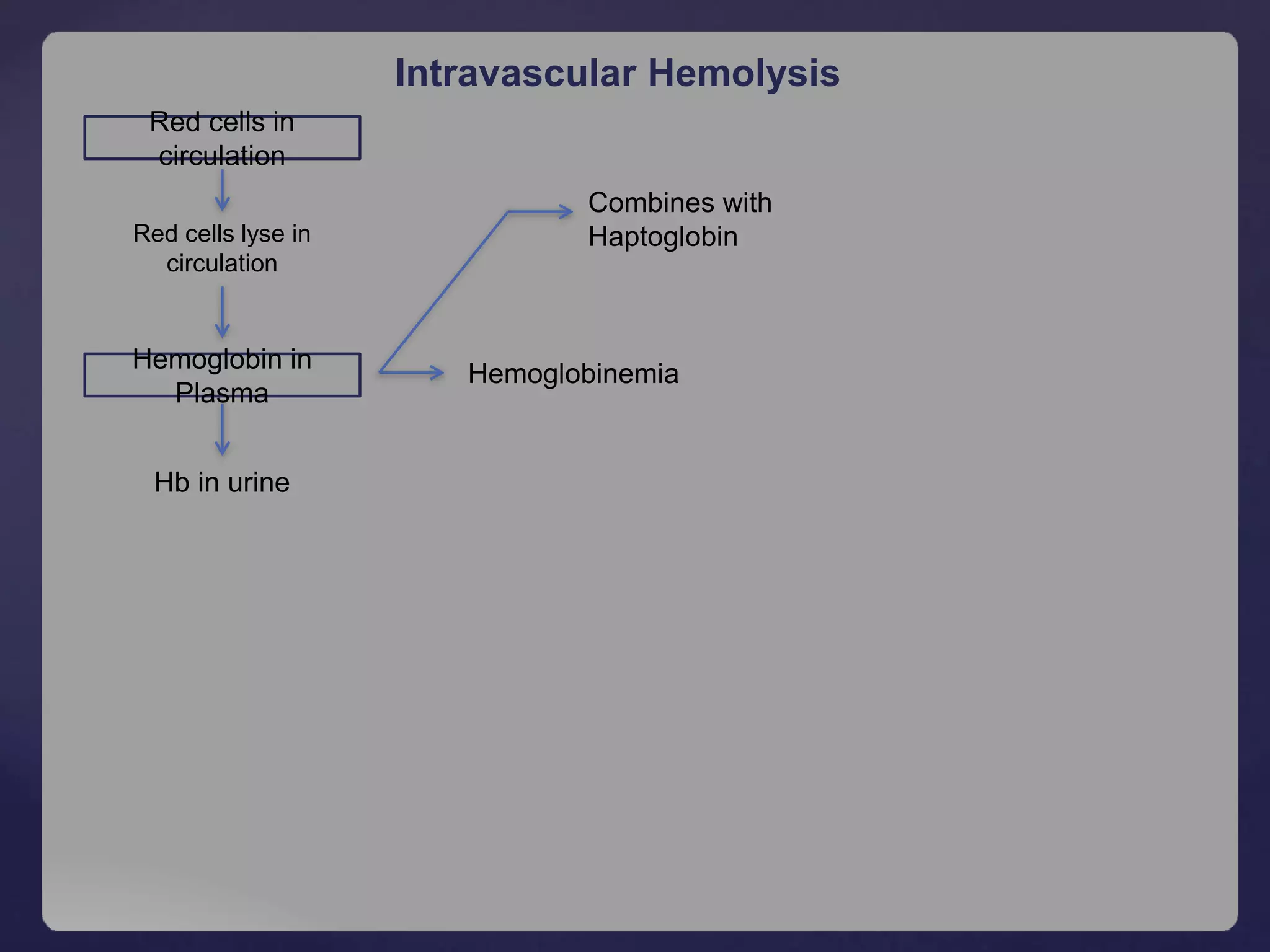
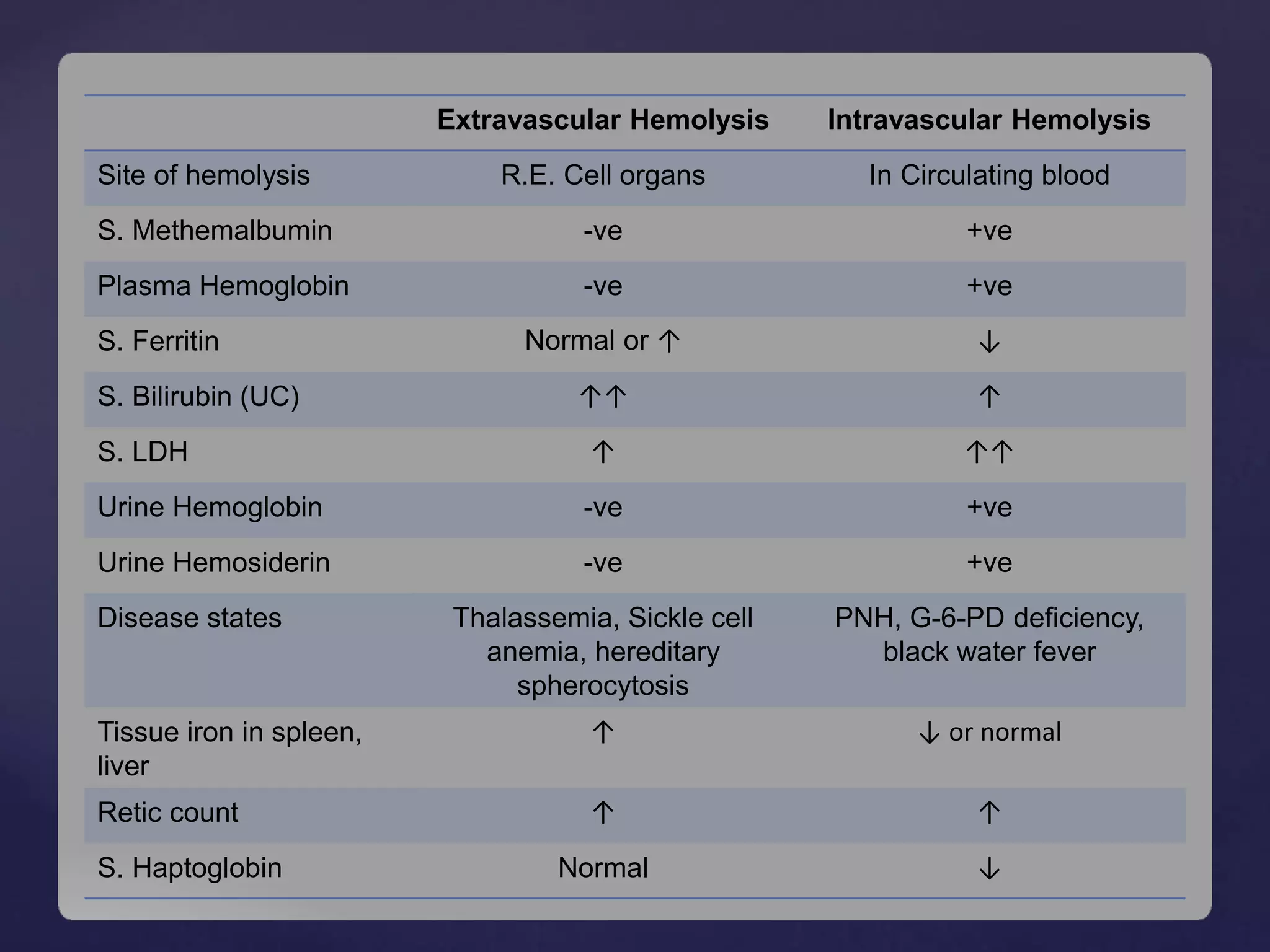
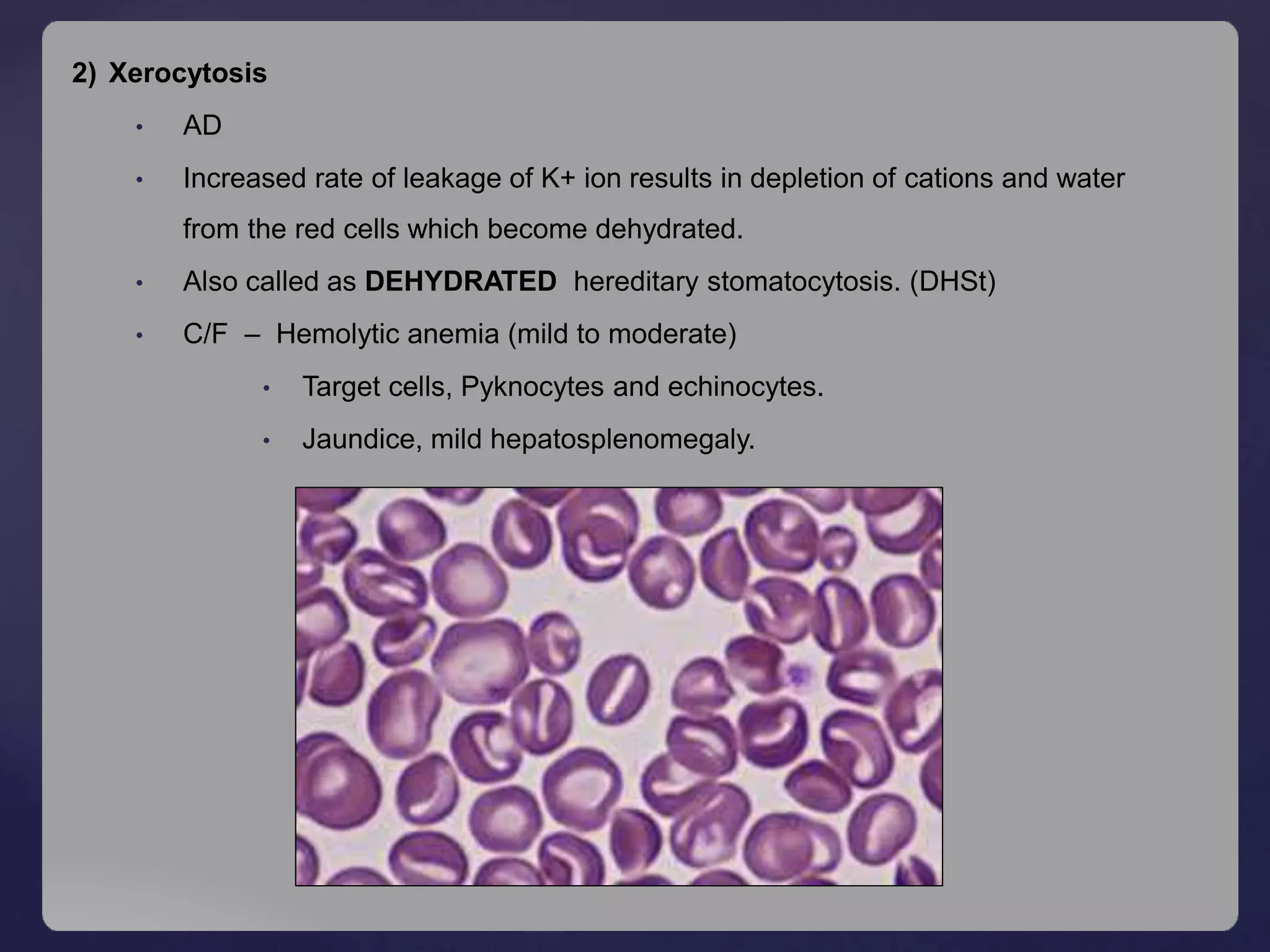
Hemolytic anemia is characterized by increased destruction of red blood cells, which can occur through extravascular or intravascular pathways. Key mechanisms include the phagocytosis of senescent red blood cells in the spleen and destruction in circulation, leading to clinical features such as anemia, jaundice, and splenomegaly. Various types of hemolytic anemia have distinct causes and diagnostic findings, with hereditary conditions like spherocytosis and acquired conditions like autoimmune hemolytic anemia being notable examples.

































![ Clinical presentation
Acute Hemolytic Anemia [self limiting], neonatal hyperbilirubinemia
Pallor, passage of dark urine
Abdominal pain, fever, chills, jaundice and severe backache
Favism, Acute renal failure
Multi organ failure
Diagnosis
Hematologic findings
Anemia with reticulocytosis
PBF - anisopoikilocytosis with polychromatophilia
Microspherocytes, bite cells, Heinz body, blister cells.
Urine – Hemoglobinuria, increased urobilinogen
Methemoglobin reduction test (MRT)
Ascorbate cyanide test
Fluorescent spot test
Cytochemical test
Dye decolorization test
Quantitative G-6-PD assay & DNA analysis by PCR](https://image.slidesharecdn.com/hemolyticanemia-180729100724/75/Hemolytic-Anemia-and-it-s-Classificaiton-37-2048.jpg)

















