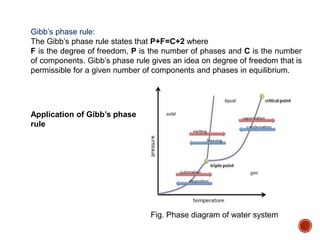
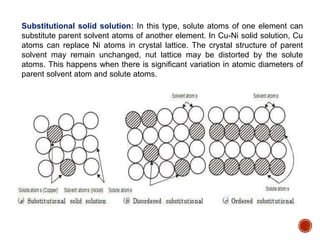
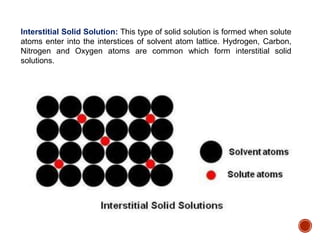
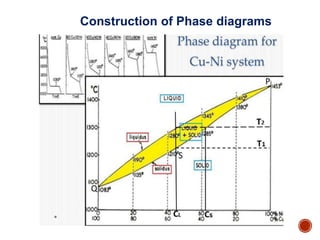
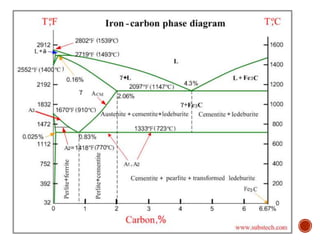
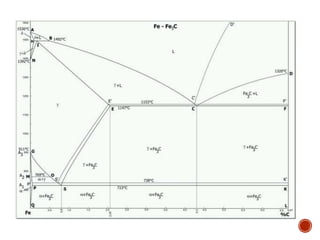
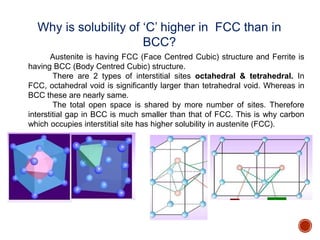
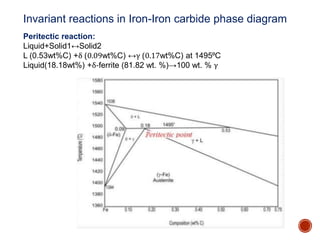
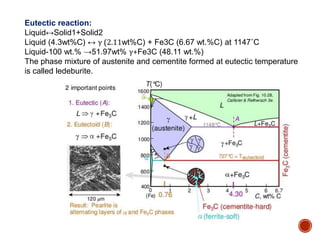
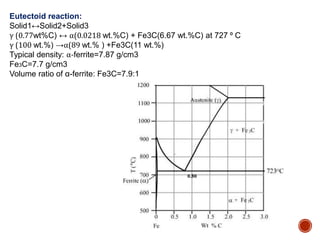
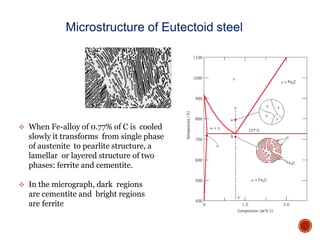
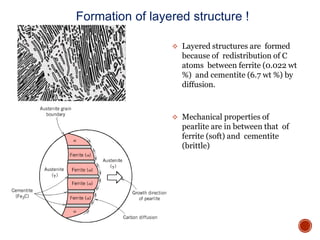
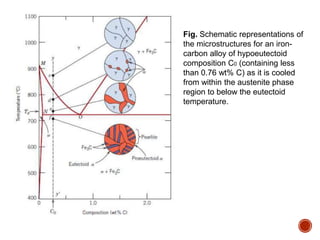
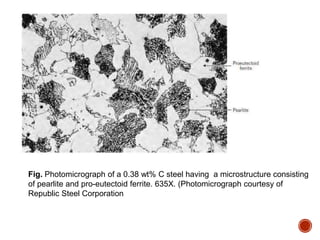
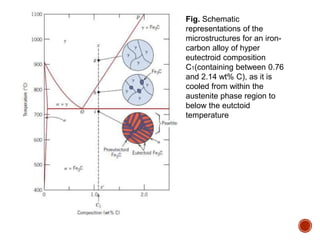
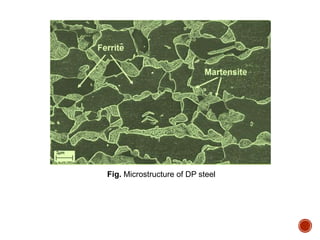
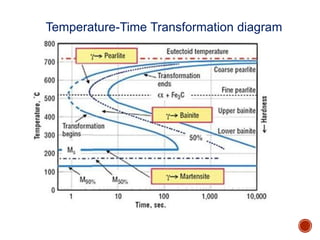
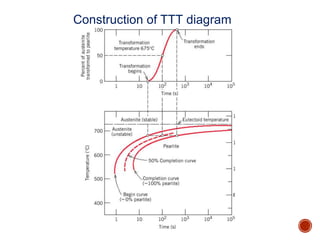
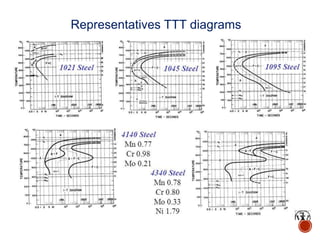
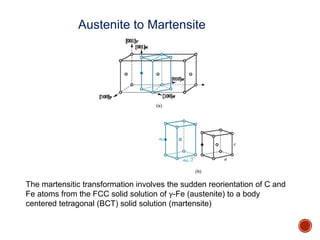
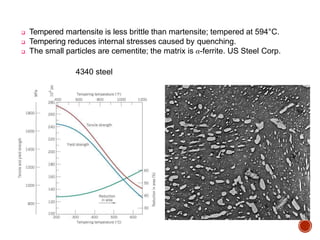
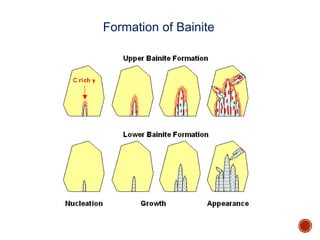
The document discusses phases, microstructures, and properties in materials. It defines a phase as a region that differs in structure and/or composition from another region. It explains that phase diagrams provide information on the number and types of phases present at different temperatures and compositions, and can show equilibrium solid solubility and temperature ranges for phase changes. Gibb's phase rule relates the number of phases, components, and degrees of freedom in a system. Solid solutions are discussed as single-phase atomic mixtures, including substitutional and interstitial types. The iron-carbon phase diagram is examined in detail, outlining the different phases such as austenite, ferrite, cementite, and eutectic or peritectic