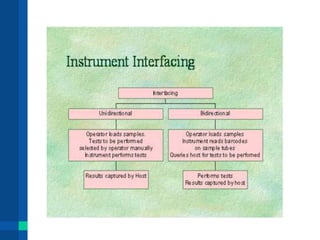
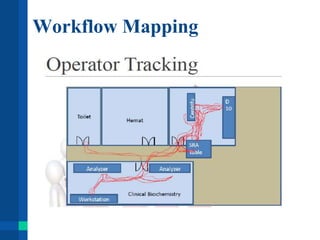
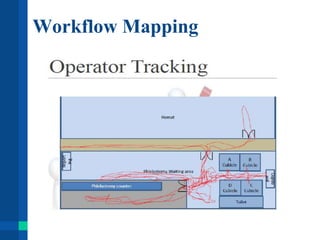
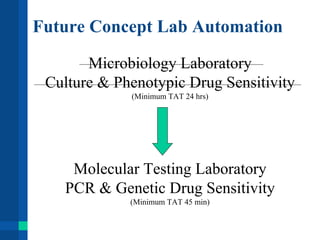
Automation in clinical laboratories can help reduce errors, decrease turnaround times, and improve productivity. It is important to fully understand current laboratory processes and identify bottlenecks before implementing automation solutions. A successful automation project requires optimizing existing workflows, clear objectives, evaluation of alternatives, and consideration of all costs. Future laboratory automation may include more integrated systems, robotics, and artificial intelligence to further improve efficiency and quality of testing.