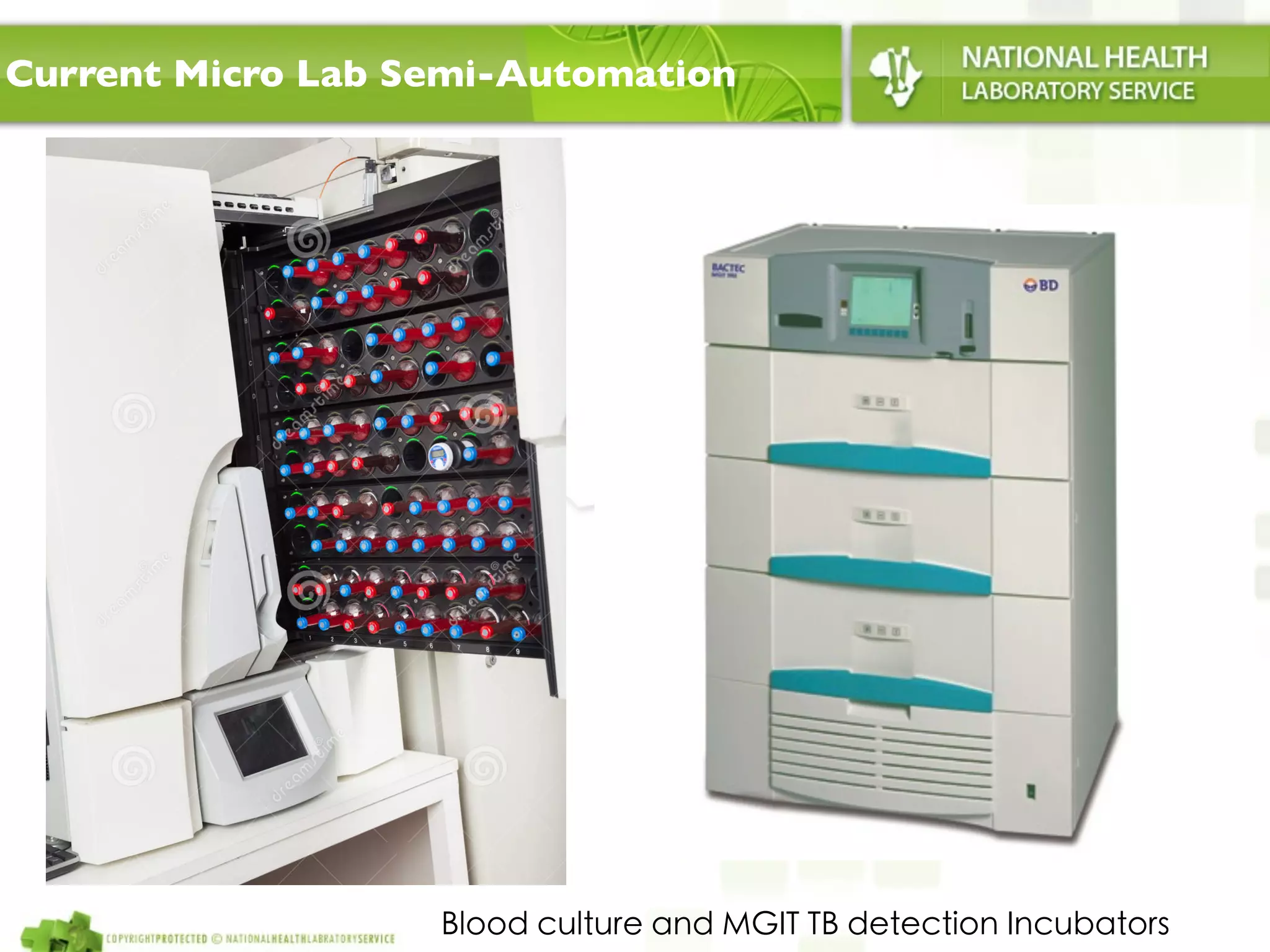
This document discusses automation in microbiology labs. It outlines several technologies currently used for semi-automation, including blood culture incubators, ID and susceptibility testing platforms, and automated stainers. Fully automated total lab automation (TLA) systems are also described, which use conveyor systems and digital imaging to automate specimen processing and plate reading. Advantages of TLA include continuous incubation, flexibility in plate reading times, efficient retrieval of plates as needed, and improved traceability. However, more research is still needed to assess the benefits of full microbiology automation.
























![SPECIMEN PROCESSORS
• Innova processor
• (BD Diagnostics, Sparks, MD)
• InoqulA full automation/manual
interaction (FA/MI) specimen-
processing device
• (BD Kiestra B.V., Drachten,
Netherlands),
• Previ Isola automated plate streaker
(bioMérieux, Inc., Hazelwood, MO)
• WASP
• Copan Diagnostics, Murrieta, CA
The recommended characteristics of
specimen processors based on studies:
accuracy,
capacity,
manufacturer’s technical support,
flexibility (specimen types, loops,
inoculation protocols, medium options,
laboratory information system [LIS] issues),
flexibility,
modularity,
and costs (initial costs, costs for any
required disposable supplies, and
operational labor costs).](https://image.slidesharecdn.com/72a01557-c567-478b-a668-985963226999-160219122000/75/Automation-in-Micro-26-2048.jpg)





