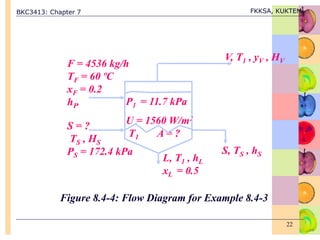
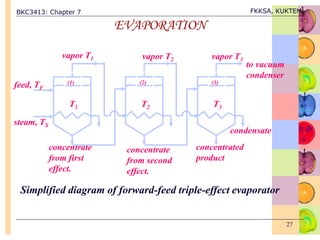
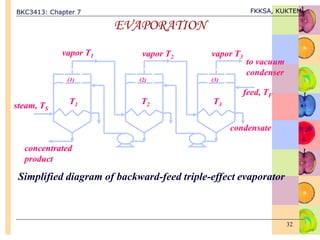
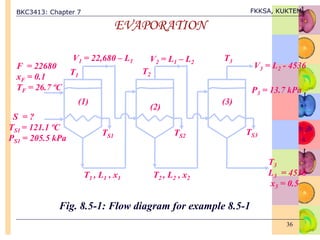
This document discusses evaporation processes and equipment. It begins by defining evaporation as adding heat to a solution to vaporize the solvent, usually water, and describes factors that affect evaporation processing like concentration, solubility, temperature, and foaming. It then covers types of evaporation equipment like single-effect and multiple-effect evaporators. For single-effect evaporators, it provides methods to calculate vapor and liquid flowrates, heat transfer area, and heat transfer coefficient. For multiple-effect evaporators, it notes they allow the latent heat of vapor from earlier effects to be recovered and reused.





![BKC3413: Chapter 7 FKKSA, KUKTEM
7
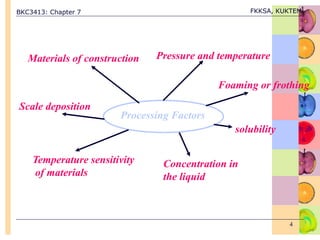
Effect of Processing Variables on Evaporator Operation.
• TF
TF < Tbp, some of latent heat of steam will be used to heat
up the cold feed, only the rest of the latent heat of steam
will be used to vaporize the feed.
Is the feed is under pressure & TF > Tbp, additional
vaporization obtained by flashing of feed.
• P1
desirable T [Q = UA(TS – T1)],
A & cost .
T1 depends on P1 will T1.](https://image.slidesharecdn.com/evaporation-221113025151-8c495e5f/85/Evaporation-ppt-7-320.jpg)



































![BKC3413: Chapter 7 FKKSA, KUKTEM
50
EVAPORATION
The average area Am = 105.0 m2 to use in
each effect.
steam economy = ???? [Q/Vapor Flowrate]
025
.
2
8960
6416
6053
5675
3
2
1
S
V
V
V](https://image.slidesharecdn.com/evaporation-221113025151-8c495e5f/85/Evaporation-ppt-50-320.jpg)
