This document discusses dose-response relationships and toxicity studies. It provides details on:
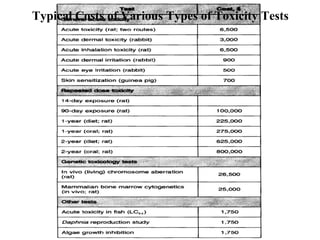
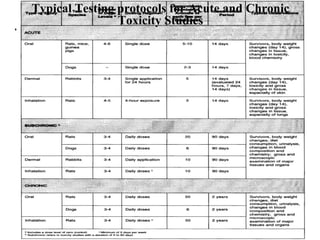
- Acute and chronic toxicity studies which determine lethal doses, target organs, and appropriate doses for further studies.
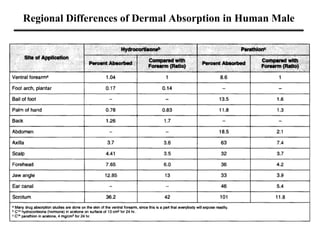
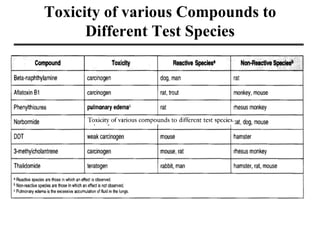
- Factors that influence toxicity like dose, exposure route, chemical properties, and individual variability in things like genetics, age and sex.
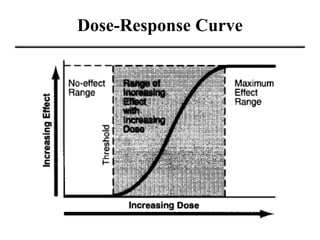
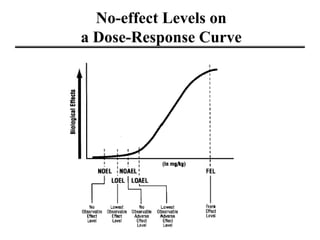
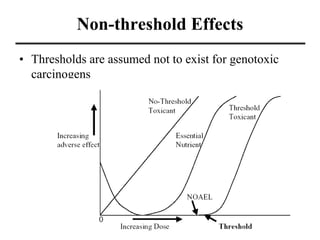
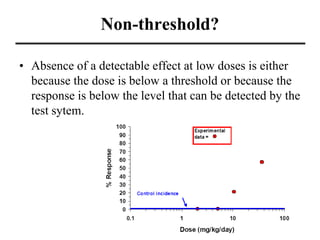
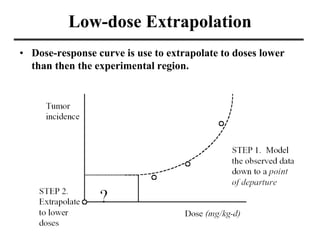
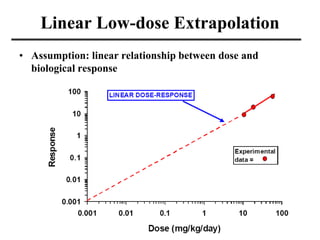
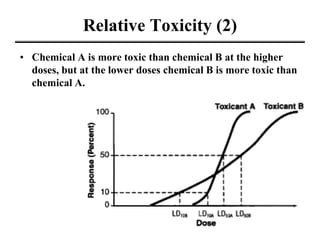
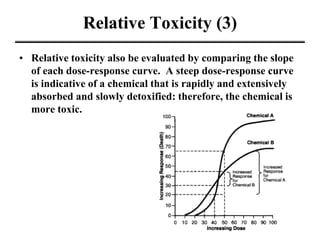
- Dose-response curves which illustrate the relationship between dose and effect and are used to determine safe exposure levels.
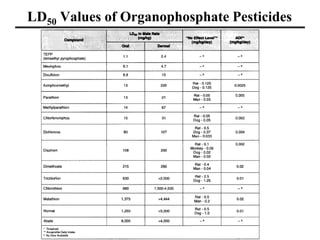
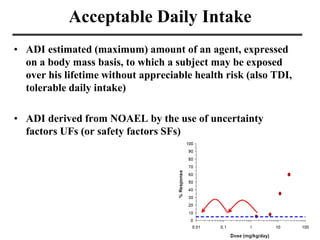
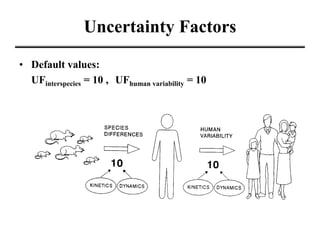
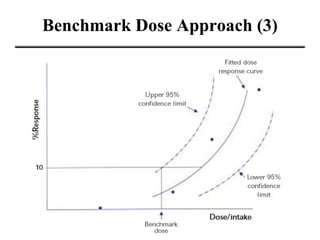
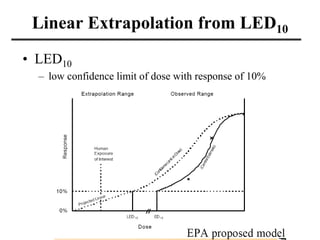
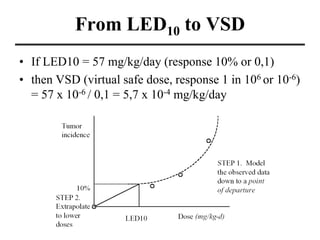
- Various terms used in toxicity assessments like NOAEL, LOAEL, BMD, RfD, and ADI.



























![Genetics
• The differences in genetics makeup both within species and
between species markedly influence the disposition and the
metabolism of toxic substances.
– For example, some individuals have an enzyme (glucose-6-
phosphate dehydrogenase) deficiency resulting in their red blood
cells being more fragile than normal. Consequently, red blood cells
of these individuals are more susceptible to chemicals such as
phenylhydrazine and primaquine, which cause hemolysis.
– The enzyme aryl hydrocarbon hydroxylase (AHH) transforms the
chemical benzo[a]pyrene to its carcinogenic metabolite. Some
individuals produce lower amounts of AHH; benzo[a]pyrene is
therefore less toxic to these individuals. Similarly, low AHH
individuals may be less likely to develop cancer from cigarette
smoke.](https://image.slidesharecdn.com/doses-230131230200-f1a7bb52/85/Doses-pdf-41-320.jpg)









![LD50 of
Chemicals
Substance Animal, Route LD50 Reference
Sucrose (table sugar) rat, oral 29,700,000,000 ng/kg [7]
Vitamin C (ascorbic acid) rat, oral 11,900,000,000 ng/kg [8]
Grain alcohol (ethanol) rat, oral 7,060,000,000 ng/kg [10]
Melamine rat, oral 6,000,000,000 ng/kg
Table Salt rat, oral 3,000,000,000 ng/kg [12]
Paracetamol (acetaminophen) rat, oral 1,944,000,000 ng/kg [13]
Metallic Arsenic rat, oral 763,000,000 ng/kg [15]
Aspirin (acetylsalicylic acid) rat, oral 200,000,000 ng/kg [17]
Caffeine rat, oral 192,000,000 ng/kg [18]
Cadmium oxide rat, oral 72,000,000 ng/kg [22]
Nicotine rat, oral 50,000,000 ng/kg [23]
Strychnine rat, oral 16,000,000 ng/kg [24]
Arsenic trioxide rat, oral 14,000,000 ng/kg [25]
Metallic Arsenic rat, intraperitoneal 13,000,000 ng/kg [26]
Sodium cyanide rat, oral 6,400,000 ng/kg [27]
Beryllium oxide rat, oral 500,000 ng/kg [30]
Aflatoxin B1 (from Aspergillus flavus) rat, oral 480,000 ng/kg [31]
Dioxin (TCDD) rat, oral 20,000 ng/kg [33]
Polonium-210 human, inhalation 10 ng/kg (estimated) [37]
Botulinum toxin (Botox)
human, oral,
injection, inhalation
1 ng/kg (estimated) [38]
Ionizing radiation human, irradiation 6 Gy](https://image.slidesharecdn.com/doses-230131230200-f1a7bb52/85/Doses-pdf-51-320.jpg)