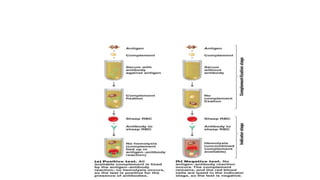
Complement fixation tests utilize complement proteins to determine the presence of antigen-antibody complexes in a sample. The test involves heating serum to remove existing complement, followed by the addition of antigens and indicators, with results indicated by hemolysis. While sensitive, this traditional method is complex, slower, and has been largely replaced by faster detection techniques like ELISA.