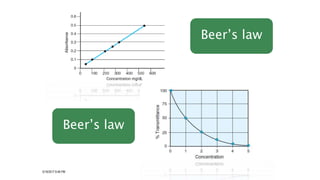
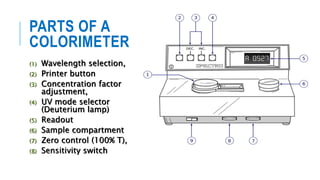
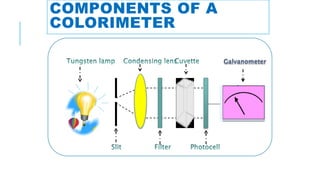
The document provides information about a colorimeter. It begins with an introduction to colorimeters, noting they measure light intensity and were invented in 1870. It then describes the basic principle that colorimeters measure light absorbed by colored solutions according to Beer's and Lambert's laws. The document outlines the typical parts of a colorimeter like its electronics and sample compartment. It discusses preparing blank, standard, and test solutions and explains Beer's and Lambert's laws. The document concludes by covering common colorimeter applications in clinical labs and industries like using them to estimate biochemical compounds in various samples.













![APPLICATIONS OF
COLORIMETER
• Estimation of biochemical compounds in
blood, plasma, serum, CSF, urine, etc.:
Glucose
Urea
Creatinine
Uric Acid
Bilirubin
Lipids
Total Proteins
Enzymes [e.g. ALT, AST, ALP]
Minerals [Calcium, Phosphorus etc.]](https://image.slidesharecdn.com/colorimeter-170519204649/85/Colorimeter-16-320.jpg)