The document provides an overview of colorimetry, an analytical technique used in clinical laboratories for biochemical estimations. It explains the principles of beer's and lambert's laws, the components and operation of a colorimeter, and its applications in various fields such as healthcare and environmental testing. Additionally, it discusses the advantages and disadvantages of using colorimeters and offers maintenance tips for effective use.

































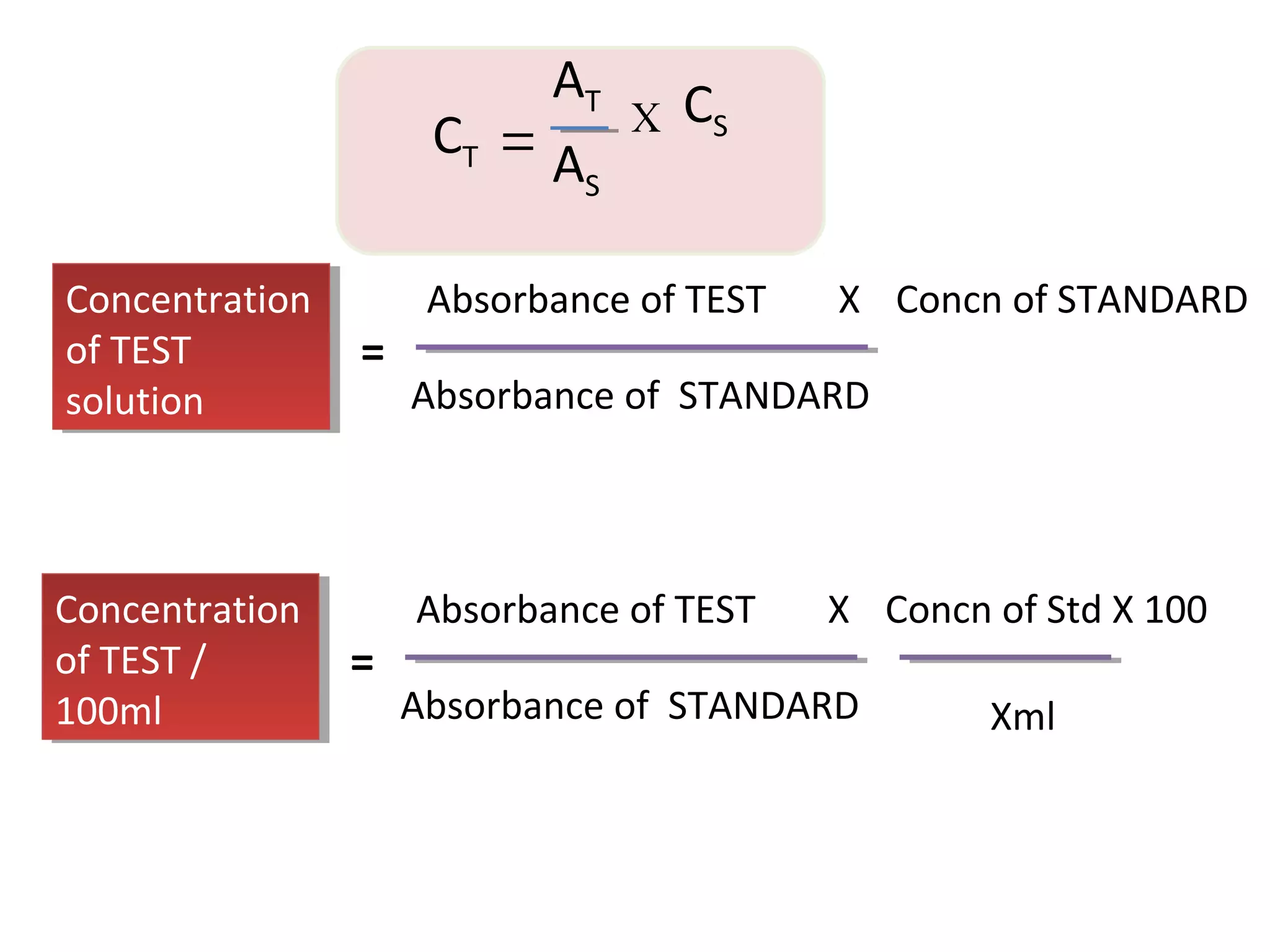



![B S1 S2 S3 S4 S5 S6 S7 T
Volume of 3% cobalt acetate
[in ml]
- 0.5 1 2 3 4 5 6 -
Volume of 1% HCl [in ml] 6 5.5 5 4 3 2 1 - -
Conc. Of cobalt acetate in
mg/6ml
- 15 30 60 90 120 150 180
O.D at 490nm 0.01 0.03 0.06 0.12 0.18 0.24 0.30 0.36 0.21](https://image.slidesharecdn.com/5-150912085108-lva1-app6892/75/Colorimeter-42-2048.jpg)










