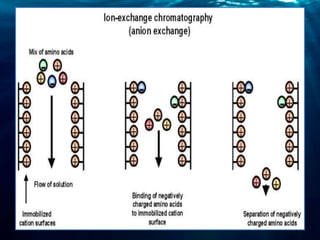
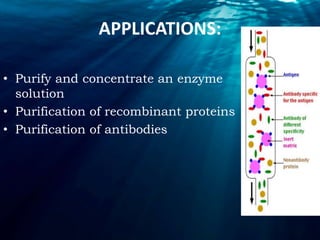
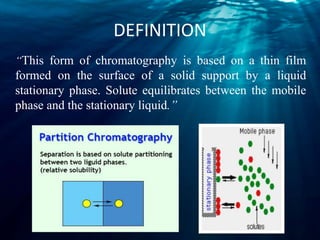
Chromatography is a technique used to separate mixtures into individual components. There are several types including thin layer, column, gas-liquid, and ion-exchange chromatography. Chromatography works by distributing components between a stationary and mobile phase. Components travel at different rates, allowing separation. Chromatography has many applications including forensic analysis, pharmaceutical purification, and research.