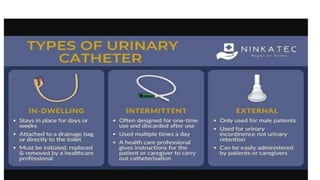
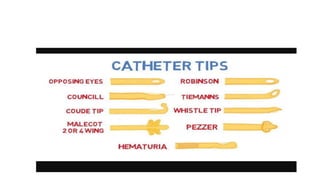
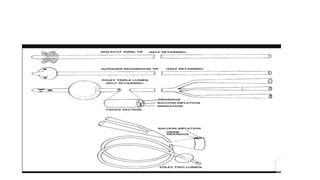
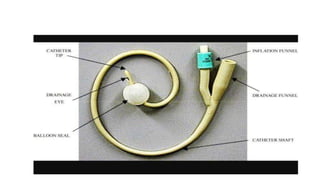
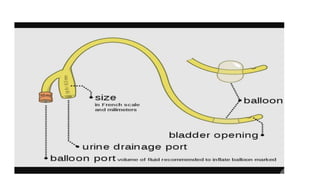
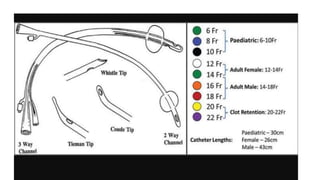
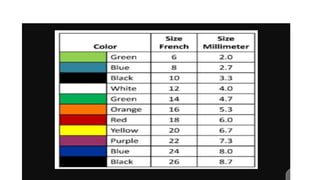
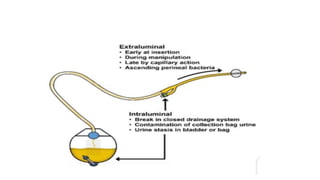
The document provides an extensive overview of urinary catheters and urethral catheterization, detailing indications, contraindications, catheter types, materials, procedures, and complications. Key highlights include the procedure's importance for various medical uses, appropriate techniques for both male and female patients, and the thorough cleaning and care required to prevent complications such as infections. It concludes that strict asepsis is crucial, emphasizing the procedure's benefits and associated risks in inexperienced hands.