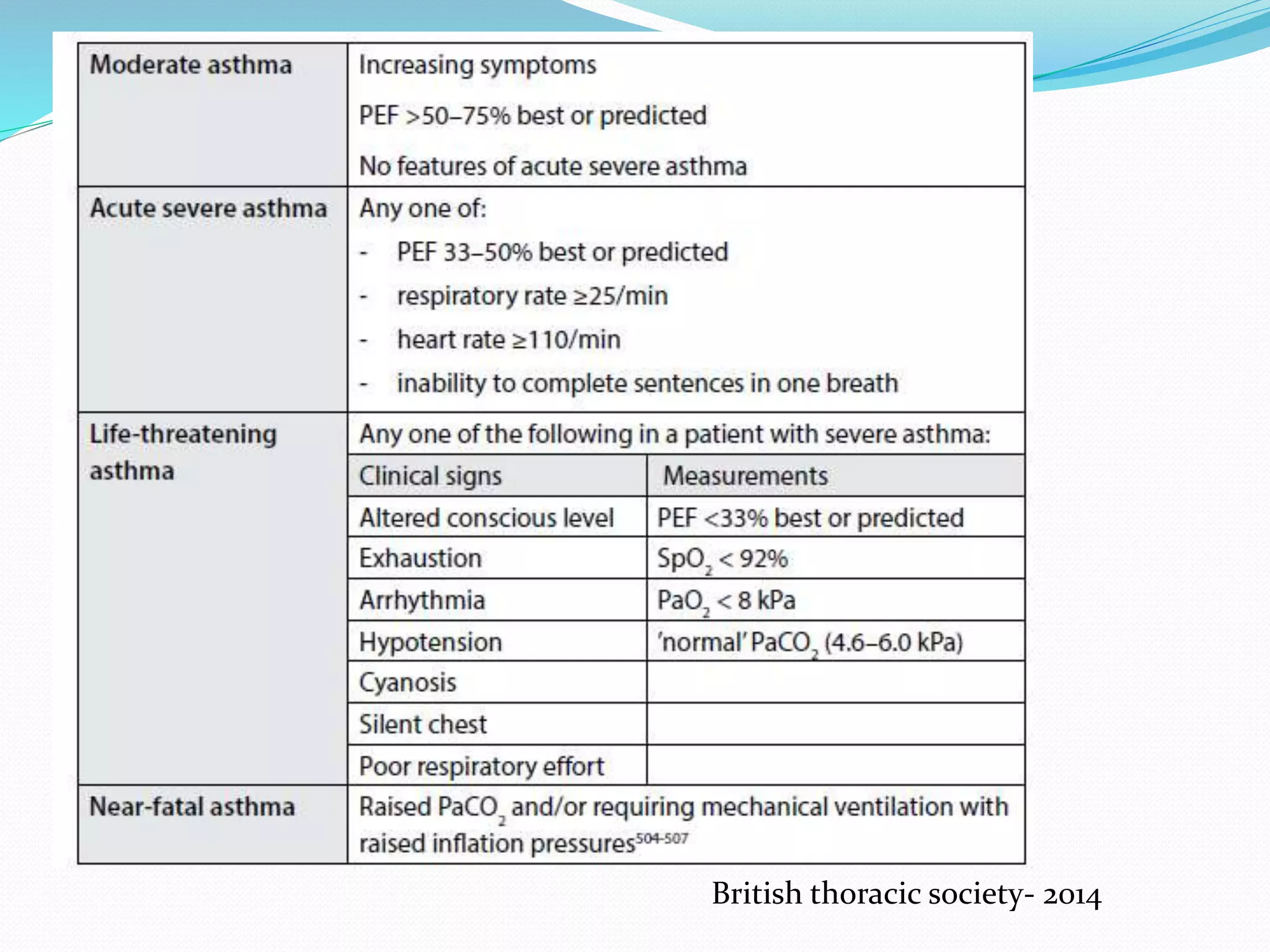
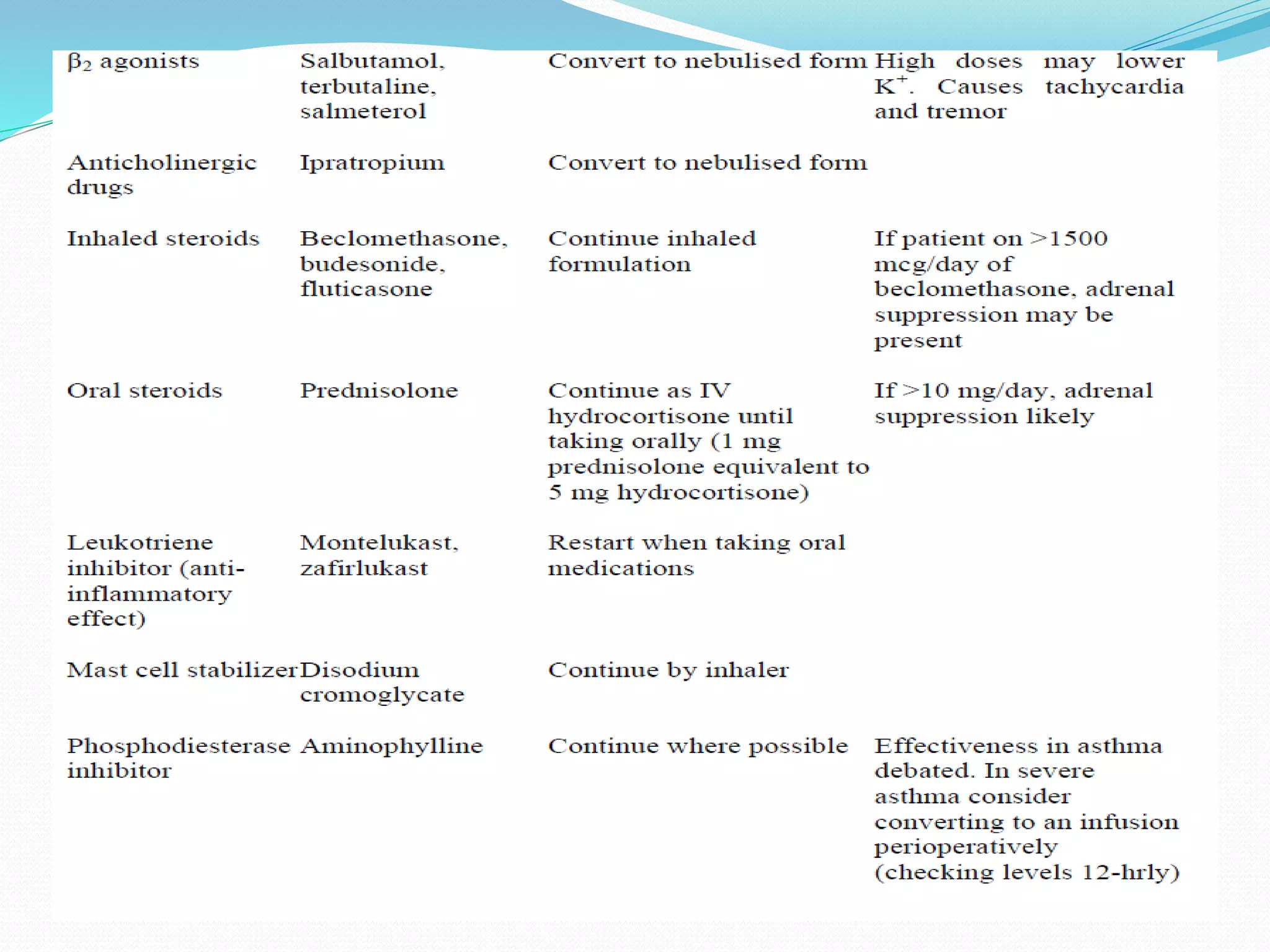
This document discusses the definition, diagnosis, treatment and management of asthma. It begins by defining asthma as a chronic inflammatory airway disease characterized by variable and recurring symptoms. It then discusses the diagnosis of asthma based on symptoms and evidence of variable airflow limitation. The document outlines treatment for acute severe asthma which focuses on relieving airflow limitation through bronchodilators and treating inflammation with corticosteroids. Key medications discussed include nebulized beta-agonists, ipratropium, systemic corticosteroids, and magnesium sulfate. Perioperative management and considerations are also reviewed.